China Approves First Brain Implant for Public Use
China just became the first country to approve a brain implant for commercial use, opening the door for people with spinal cord injuries to regain movement. While similar devices have existed in research labs for decades, this marks a breakthrough in making the technology accessible beyond clinical trials.
For the first time in history, a brain implant has been approved for commercial use outside of research trials.
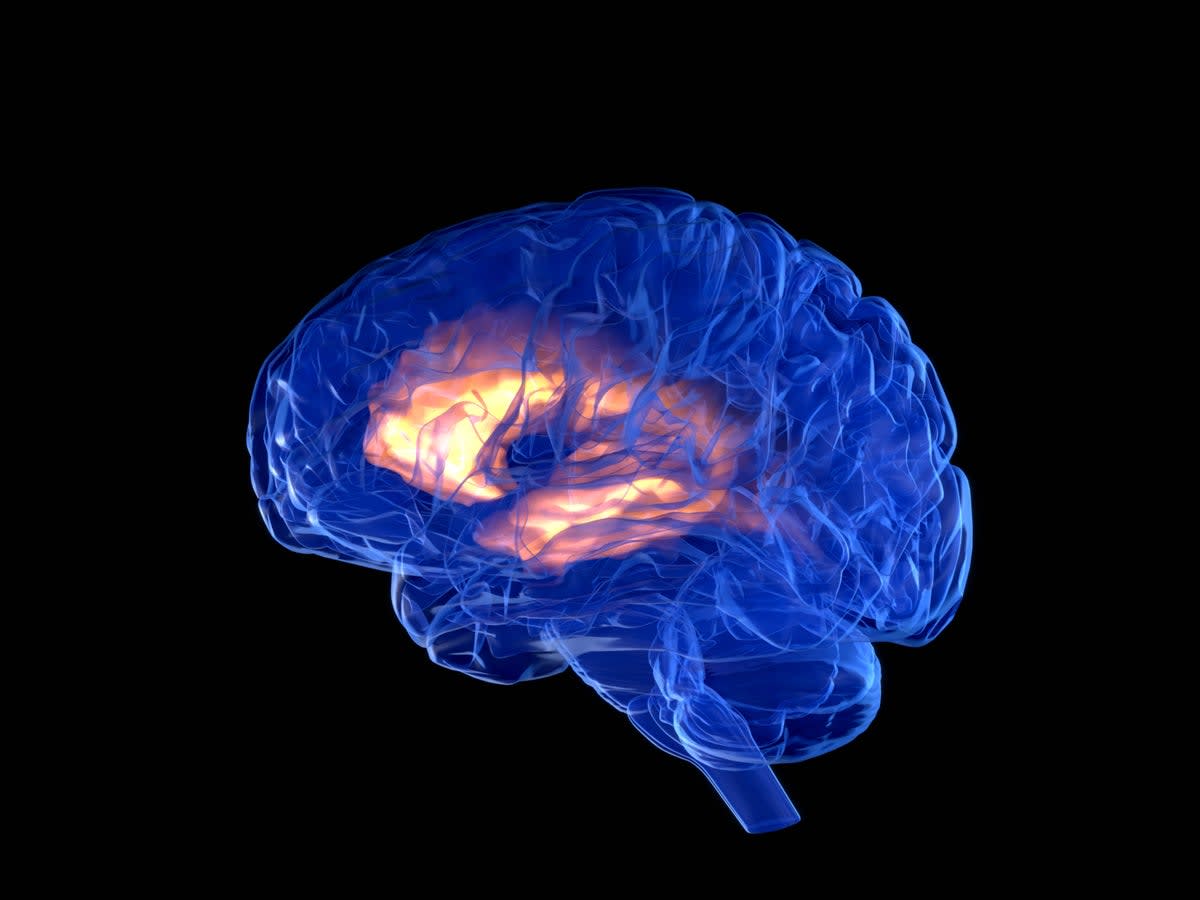
China greenlit Shanghai-based Neuracle Medical Technology's brain-computer interface (BCI) for people with partial spinal cord injuries. The coin-sized wireless implant sits on the brain's surface and controls a robotic glove, helping users regain upper body function.
Brain-computer interfaces work by recording electrical signals from neurons and translating them into commands. The software decodes these signals so users can control prosthetic limbs, computer cursors, or in this case, a robotic glove that assists with daily tasks.
While companies like Elon Musk's Neuralink have made headlines, they're still in the experimental phase. In the United States, no BCI has been approved for commercial use yet. Neuralink has enrolled 21 participants in trials as of January 2026, and other startups like Synchron and Paradromics are also testing their devices.
The technology itself isn't new. The BrainGate consortium created one of the first BCIs in the early 2000s, enabling people with paralysis from conditions like ALS or locked-in syndrome to control computer mice and type on virtual keyboards. What's revolutionary here is the leap from research to real-world availability.

The Ripple Effect
This approval signals a major shift in how we treat paralysis and spinal cord injuries. For decades, people have participated in clinical trials with hope but no guarantee of access. Now, patients in China with qualifying injuries can actually get this technology.
The move may accelerate approvals worldwide. When one country demonstrates safety and effectiveness at scale, regulatory agencies elsewhere often follow. American and European companies developing similar devices now have a roadmap for commercialization.
Of course, brain surgery carries risks including infection and complications. The implants can also move over time or cause scar tissue that weakens their signals. But for people living with paralysis, the potential to regain independence often outweighs those concerns.
Neuracle's device targets people who still have some upper arm function, making it practical for those who might benefit most from enhanced mobility. It's a careful, measured approach to introducing transformative technology.
This isn't science fiction anymore—it's medical reality, and it's giving people their lives back.
More Images


Based on reporting by Scientific American
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


