FDA-Approved Drug May Block Breast Cancer Spread
Scientists discovered an existing FDA-approved medication could prevent the deadliest part of breast cancer: when it spreads to other organs. The breakthrough offers new hope for patients with aggressive triple negative breast cancer.
A drug already sitting on pharmacy shelves might hold the key to stopping breast cancer before it turns deadly.
Researchers at Dana-Farber Cancer Institute found that mifepristone, a medication already approved by the FDA, can prevent breast cancer cells from spreading to other organs in animal studies. The discovery could transform how doctors fight triple negative breast cancer, the most aggressive form of the disease.
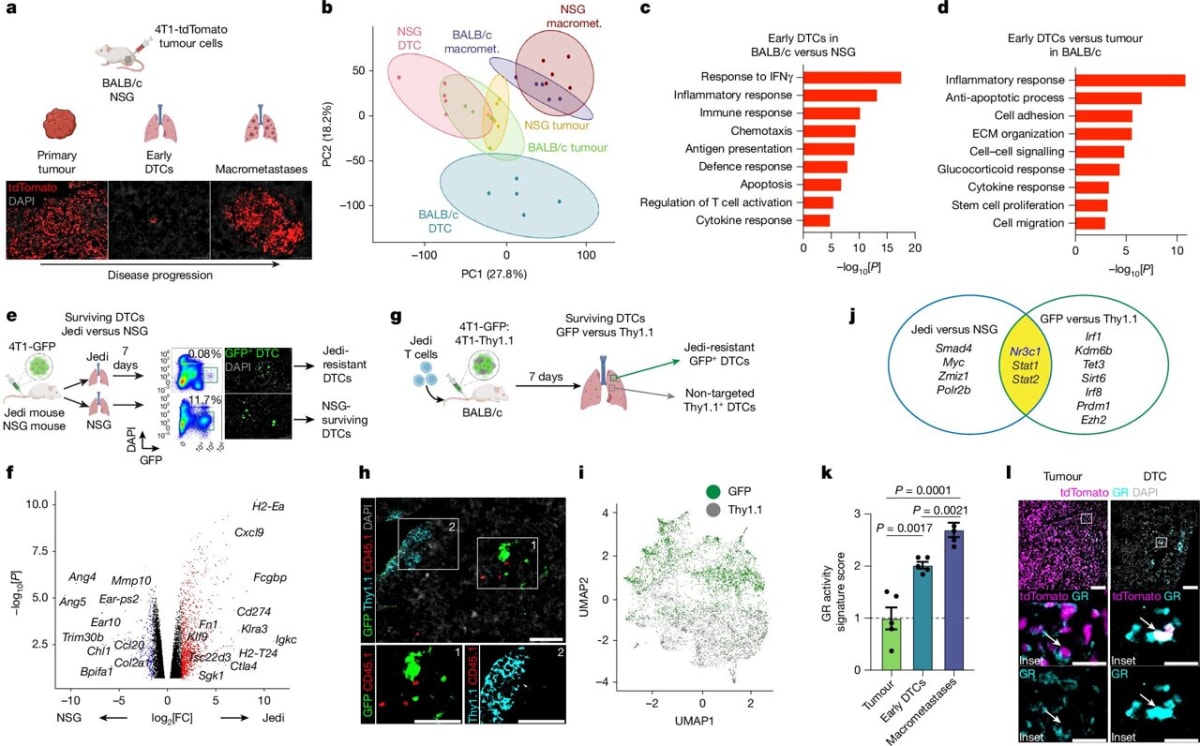
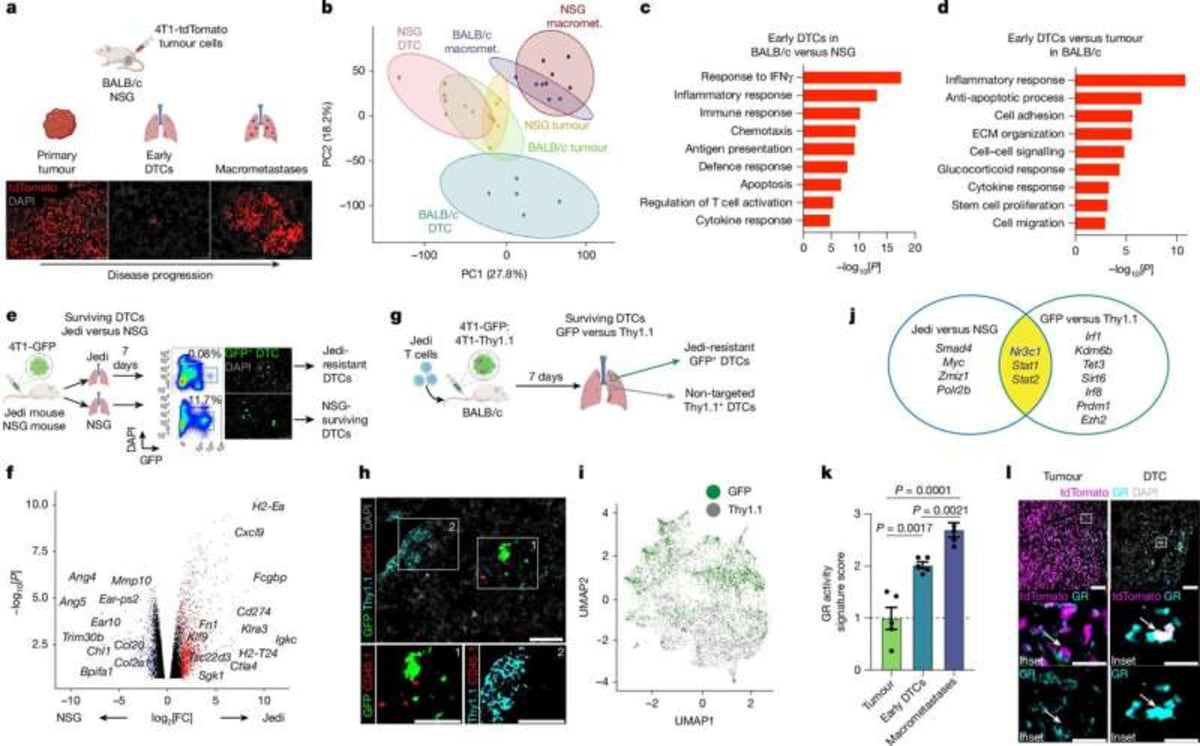
Most cancer deaths happen not from the original tumor, but when cancer cells break away and grow in distant organs like the lungs, liver, or brain. Dr. Judith Agudo and her team wanted to understand how these traveling cancer cells manage to hide from the immune system.
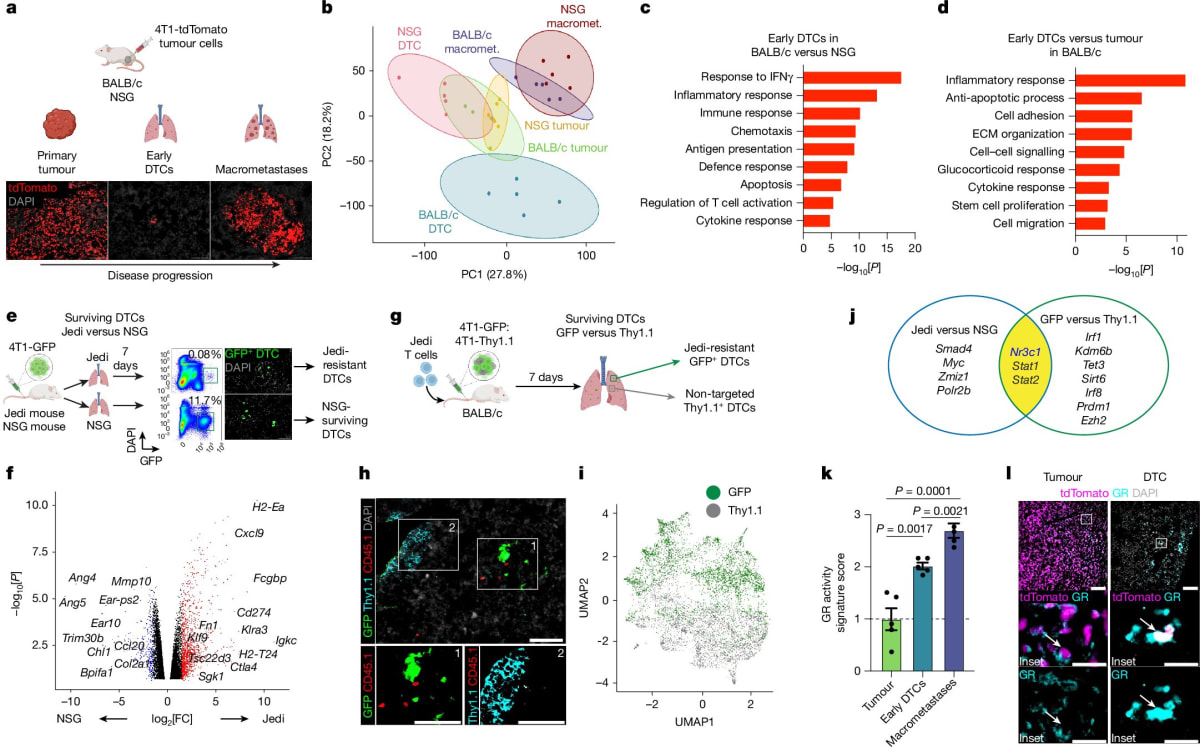
The team invented a new tracking tool called Jedi to hunt down escaped cancer cells in mice. They discovered something surprising: some tumor cells activate a protective shield using the glucocorticoid receptor, which helps them evade immune attack in their new locations.
That's where mifepristone comes in. The drug blocks this receptor, stripping away the cancer cells' protective shield and allowing the immune system to destroy them. In mice, the treatment reduced the number of new cancer sites and worked even better when combined with existing immunotherapy drugs.

The combination therapy not only shrunk metastases but also extended survival in the animal models. While human trials haven't started yet, previous research shows this same glucocorticoid receptor activity appears in human triple negative breast cancer samples and links to worse outcomes.
Why This Inspires
This research flips the script on cancer treatment. Instead of only fighting the tumor we can see, scientists are learning to stop cancer cells during their most vulnerable moment: when they're traveling to new organs.
The fact that mifepristone is already FDA-approved for other uses means it could potentially move to human trials faster than a brand new drug. That timeline matters enormously for patients facing aggressive cancers.
Triple negative breast cancer affects thousands of women each year and responds poorly to many standard treatments. This approach offers a completely new angle, one that harnesses the body's own immune system as the weapon.
If the strategy works in humans, it could extend beyond breast cancer to other solid tumors like prostate and colon cancer, where metastasis remains the biggest threat to survival.
Scientists are turning cancer's own escape route into a trap.
More Images


Based on reporting by Medical Xpress
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it