
FDA Approves Brain Implant Trial for Severe Depression
A tiny brain implant that treats depression from home just won FDA approval for human trials. The device could help millions who haven't found relief from traditional medications.
Nearly 3 million Americans live with treatment-resistant depression, and a breakthrough device might finally offer them hope.
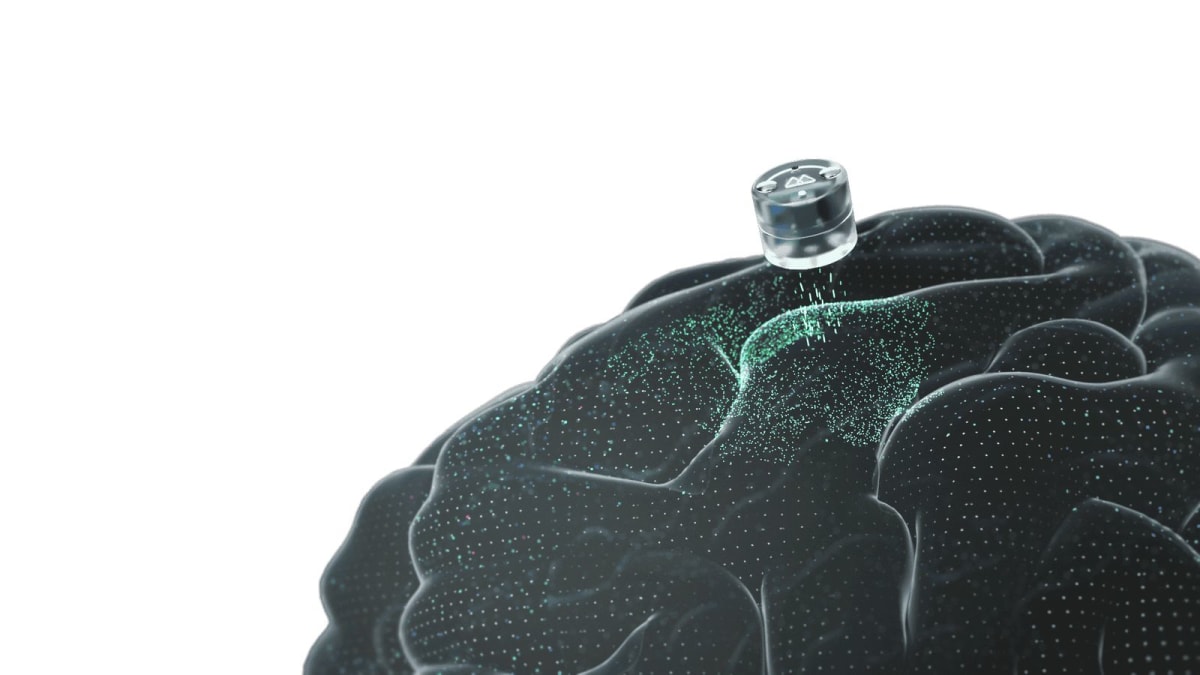
Houston startup Motif Neurotech just received FDA approval to test a blueberry-sized brain implant that treats severe depression. Unlike existing treatments that require weeks of daily hospital visits, this device lets patients receive therapy at home using a special baseball cap.
The implant sits just above the brain's protective membrane, targeting an area that controls high-level thinking and motivation. In people with major depression, this network runs too slow. The device sends specific electrical pulses to wake it up, strengthening the brain's natural connections over time.
"We think we can drive that neuroplasticity that creates stronger connectivity for patients with depression, so that they can get out of bed in the morning, call their friends, go to the gym," says Jacob Robinson, Motif's CEO and cofounder.
The procedure takes just 20 minutes in an outpatient setting. No brain surgery required. Patients charge the implant by wearing a baseball cap for 10 to 20 minutes several times daily during the first 10 days. After that, they switch to occasional maintenance doses.

Early results could show people entering remission within those first 10 days. That's dramatically faster than current FDA-approved options like transcranial magnetic stimulation, which requires five treatments weekly for six weeks.
The yearlong trial will track about 10 participants, monitoring both safety and whether depression symptoms actually improve. Researchers will also measure quality of life, anxiety levels, and cognitive function.
The Ripple Effect
Beyond treating depression, this technology could transform psychiatry itself. Future versions will record brain activity over time, giving doctors objective data about mental health for the first time.
Robinson compares current psychiatry to managing diabetes based purely on how someone feels. Brain monitoring would be like finally having a blood sugar meter. Doctors could personalize treatments based on what's actually happening in the brain rather than relying on patient self-reports alone.
The device joins a growing wave of brain-computer interfaces, though most others focus on helping paralyzed people communicate. Motif chose a different path: making cutting-edge neurotechnology accessible for one of the most common mental health conditions.
For people who've tried medication after medication without relief, this trial represents something they may have lost: the possibility that tomorrow could feel different.
More Images



Based on reporting by Wired
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


