FDA Fast-Tracks Gene Therapy for Rare Diseases
The FDA just made it easier for millions of Americans with rare diseases to access life-saving gene therapies without waiting years for traditional clinical trials. Families who were told to "wait for science to catch up" now have real hope.
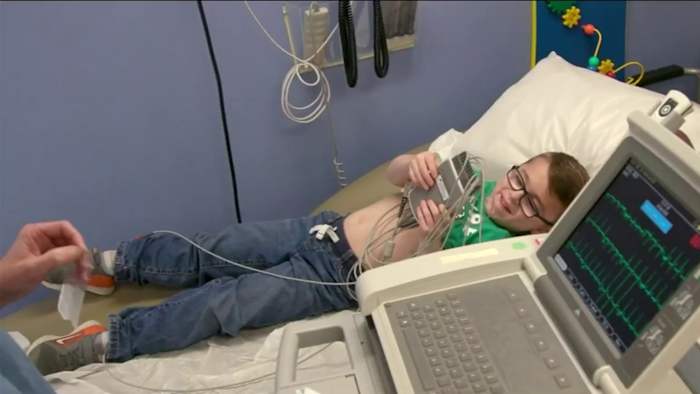
For families watching their children battle ultra-rare genetic diseases, the answer has always been the same: not enough patients, trials take too long, maybe someday. That changes now.
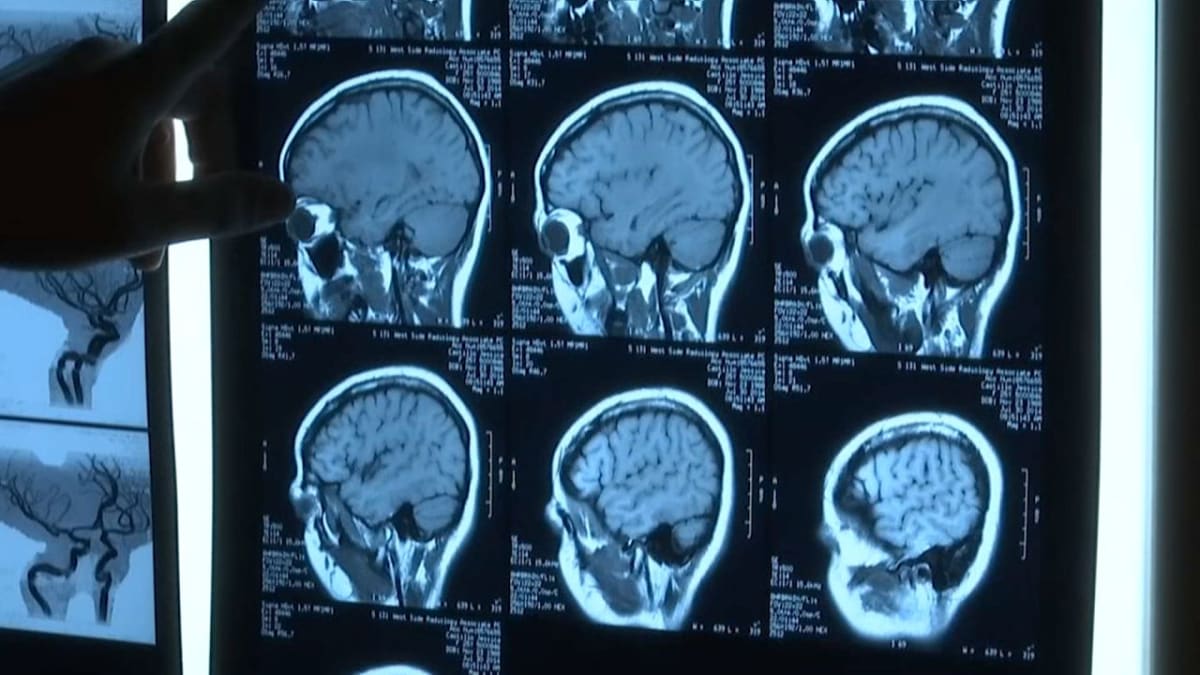
The FDA announced Monday a groundbreaking policy that lets doctors use gene-editing treatments for rare diseases based on how the therapy should work, not years of traditional studies. For conditions so rare that gathering enough patients for a trial is nearly impossible, this opens a door that was previously locked.
The policy change started with one Pennsylvania baby. Doctors at Children's Hospital of Philadelphia saved an infant born with a devastating genetic liver disorder by creating a custom gene-editing treatment targeting his specific mutation. They realized they could repeat this process for other children with similar conditions without starting from square one each time.
"For decades families heard the same thing: There are not enough patients. The approval will take too long," said Health and Human Services Secretary Robert F. Kennedy Jr. "That ends today."
About 30 million Americans live with a rare disease. Thousands of these conditions could potentially be cured through gene-editing technology like CRISPR, which scientists are already using to treat sickle cell disease and studying for cancer, inherited high cholesterol, and genetic blindness.

The challenge has been economics and red tape. Pharmaceutical companies haven't had strong incentives to develop treatments for extremely rare disorders, and each case requiring its own lengthy approval process made personalized medicine impractical.
The Ripple Effect
This new approach creates a template system. When doctors develop a gene-editing treatment for one rare genetic condition, they can adapt it for similar disorders without repeating the entire regulatory process. That makes treatments more affordable and accessible.
Dr. Kiran Musunuru, who helped develop the treatment for the Pennsylvania baby, explained the potential: "We realized we can do this over and over again, individualizing the therapy for many patients."
Scientists across the country are celebrating. "To hear HHS leadership say a disease with 100 causing mutations will no longer require 100 clinical trials sounds like a veritable Ode to Joy," wrote geneticist Fyodor Urnov of UC Berkeley. "It means we will be able to treat children faster and more affordably."
The policy applies beyond gene-editing to other advanced technologies that could treat rare conditions. While some experts expressed concern about ensuring the pathway stays focused on truly rare diseases where traditional trials aren't feasible, the overall response has been enthusiastic.
Doctors who have watched families struggle now see a path forward that seemed impossible just months ago.
More Images


Based on reporting by Google News - New Treatment
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it