First Brain-Crossing Hunter Syndrome Drug Wins FDA Approval
After 20 years without new treatments, the FDA approved Avlayah, the first therapy designed to treat both physical and neurological symptoms of Hunter syndrome. The breakthrough offers hope to families battling this rare disease that causes progressive organ damage and early death.
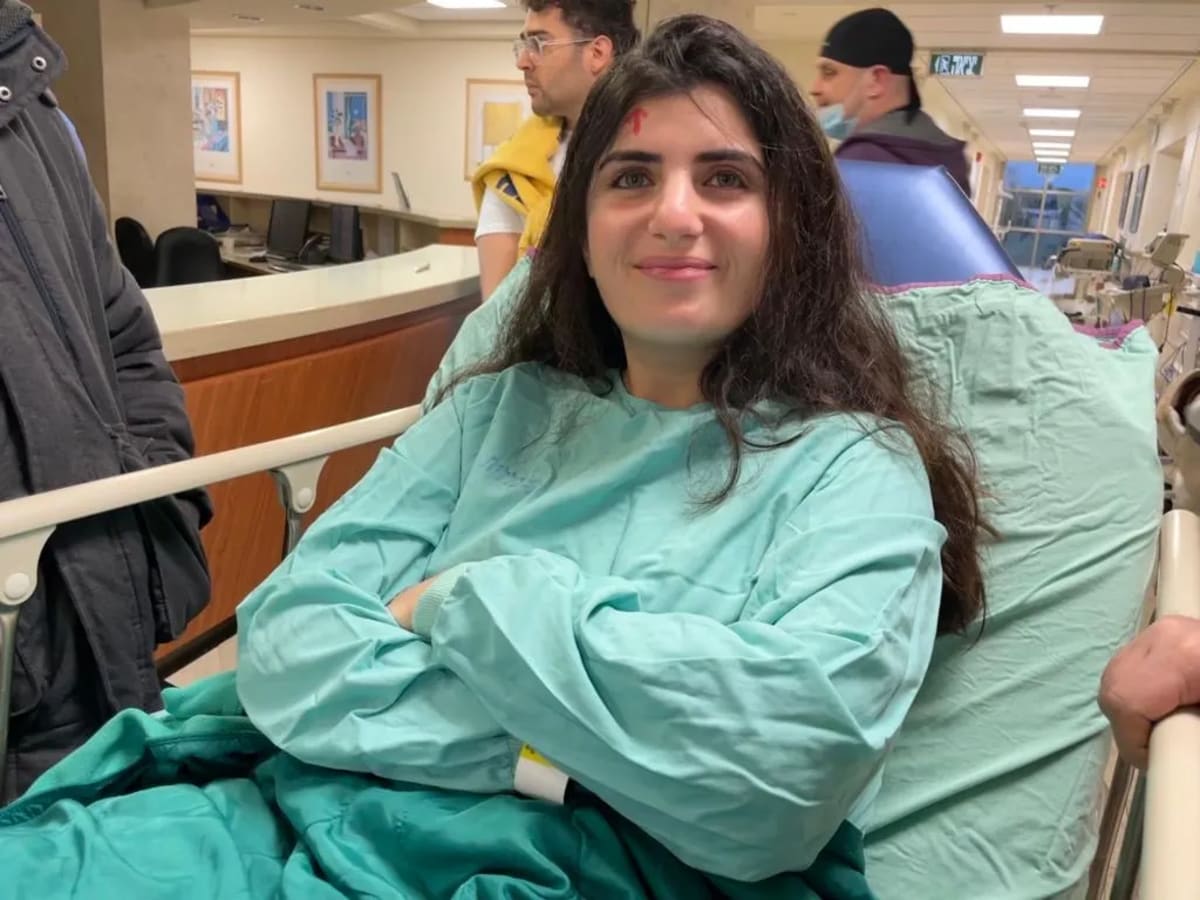
Families fighting Hunter syndrome finally have a reason to celebrate after two decades of waiting for medical progress.
On March 25, the FDA granted accelerated approval for Avlayah, a groundbreaking enzyme replacement therapy created by Denali Therapeutics. It's the first drug designed to cross the blood-brain barrier and treat both the neurological and physical symptoms of this devastating rare disease.
Hunter syndrome affects the body's ability to break down certain sugars, causing toxic buildup that damages multiple organs over time. Since 2006, the only FDA-approved treatment couldn't reach the brain, leaving patients to suffer progressive neurological decline despite therapy.
For the small but determined Hunter syndrome community, this approval represents genuine scientific innovation. Avlayah works differently than existing treatments, finally offering the possibility of addressing the neurological symptoms that have stolen so much from patients and their families.
The approval comes as advocacy groups like Project Alive and Siblings with a Mission have pushed for better treatments. Their efforts highlight how patient communities can drive medical progress even for rare diseases affecting small populations.

The Bright Side
While the current FDA label limits Avlayah to patients under 16, Denali Therapeutics is already conducting expanded trials including participants up to age 26. This signals confidence that the benefits extend beyond the initial approval, with more access likely coming as research continues.
The scientific reasoning behind the drug suggests it should work for adults too, since the disease progresses through the same mechanism regardless of age. Insurance coverage challenges exist for now, but this first approval creates a pathway forward that didn't exist before.
For researchers studying rare diseases, Avlayah's approval demonstrates that crossing the blood-brain barrier is possible. This breakthrough could accelerate similar treatments for other conditions affecting the brain, potentially helping thousands more families facing different rare diseases.
Patient advocacy played a crucial role in getting this therapy developed and approved faster. When families share their stories and push for solutions, pharmaceutical companies and regulators listen, creating momentum that saves lives.
The phase 2/3 trials currently underway will provide more data to support expanded access, potentially removing age restrictions as evidence builds. Each trial participant contributes to knowledge that could help the entire community gain better treatment options.
After watching loved ones gradually lose abilities to a disease with no good options, Hunter syndrome families now have something they haven't had in 20 years: a genuinely new tool in the fight, and solid reasons to hope for continued progress ahead.
More Images



Based on reporting by STAT News
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it