Magnetic Brain Pulses May Treat Postpartum Depression
A groundbreaking trial is testing whether magnetic brain stimulation could safely treat postpartum depression without medication, offering hope to new moms who can't or won't take antidepressants while breastfeeding. The drug-free treatment takes just five days and could expand options for the 1 in 8 women who experience this condition.
New mothers struggling with postpartum depression may soon have a safe, medication-free treatment option that takes less than a week to complete.
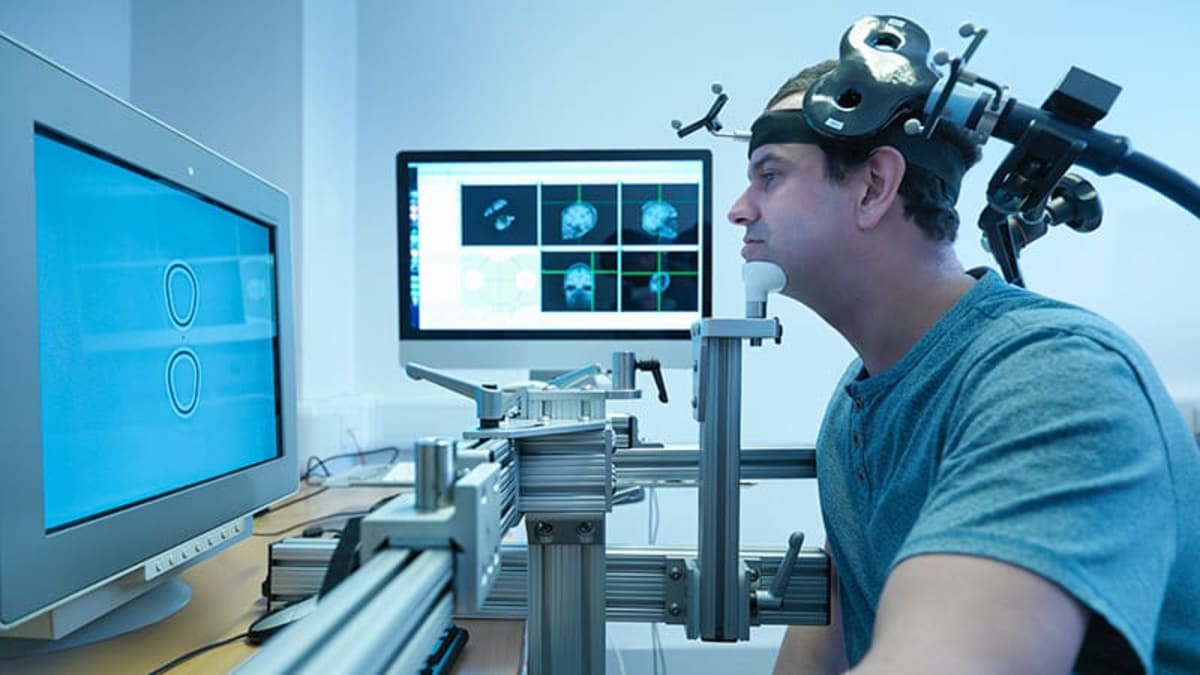
Researchers are testing repetitive transcranial magnetic stimulation on 192 women with postpartum depression in a groundbreaking trial. The treatment uses magnetic pulses to stimulate specific brain regions associated with depression, offering hope to moms who avoid medication while breastfeeding.
"Having effective treatments for postpartum depression has incredible public health consequences for new moms, their children, and their families," said Dr. Lee Cohen, director of the Ammon-Pinizzotto Center for Women's Mental Health at Mass General Hospital.
The treatment addresses a critical gap in care. About 1 in 8 American women who give birth report symptoms of postpartum depression, yet many won't take antidepressants while nursing, and others stop their medications as soon as they become pregnant.
During the trial, participants receive 10 sessions per day over five consecutive days at an outpatient clinic. A coil secured to the scalp delivers 90,000 magnetic pulses to the brain's prefrontal cortex, targeting what researchers call the "sadness center."
After childbirth, plummeting hormones and exhaustion can trigger intense negative emotions. The brain's prefrontal cortex struggles to regulate these feelings, creating the perfect storm for depression. The magnetic stimulation helps the prefrontal cortex regain control over the brain regions processing sadness and grief.

The FDA already approves this stimulation for major depressive disorder and treatment-resistant depression, where it helps about 40% of patients. Trial leader Dr. Martijn Figee of Mount Sinai believes it could trigger a "biological reset" for new moms.
The Ripple Effect
This research arrives at a crucial moment. The first FDA-approved drug specifically for postpartum depression only came in 2019, and it required a 60-hour IV infusion costing nearly $40,000. An oral medication followed in 2023, but options remain limited for a condition affecting hundreds of thousands of women each year.
Other teams are exploring even more accessible solutions. Some researchers are studying whether moms could use portable devices for similar brain stimulation at home, eliminating the challenge of leaving a newborn to visit a clinic five days straight.
"We are trying to facilitate easier access to these treatments so people don't have to come into the office all day for 5 days," Figee said.
The trial will collect data through 2029. If successful, the team will seek expanded FDA approval, potentially making the treatment covered by insurance and widely available.
Beyond just treating depression, researchers are also investigating whether the stimulation helps moms with anxiety or obsessive-compulsive disorder, which often occur alongside postpartum depression. The growing recognition of postpartum depression as a serious public health issue is driving decreased stigma and increased research funding.
New mothers deserve every possible tool to help them through one of life's most challenging transitions, and science is finally catching up.
Based on reporting by Google News - New Treatment
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it