New Tool Cuts Clinical Trial Delays by 95M Patient Records
A digital health company just made it dramatically easier for medical researchers to find the right patients for clinical trials, potentially speeding up the arrival of new treatments. Real-world health data from 95 million patients is now helping researchers avoid the delays that have plagued medical studies for decades.
Clinical trials have long struggled with a frustrating problem: finding the right patients takes too long, and many research sites enroll too few people to be useful. Now a digital health company has launched tools that could transform how new medical treatments reach patients who need them.
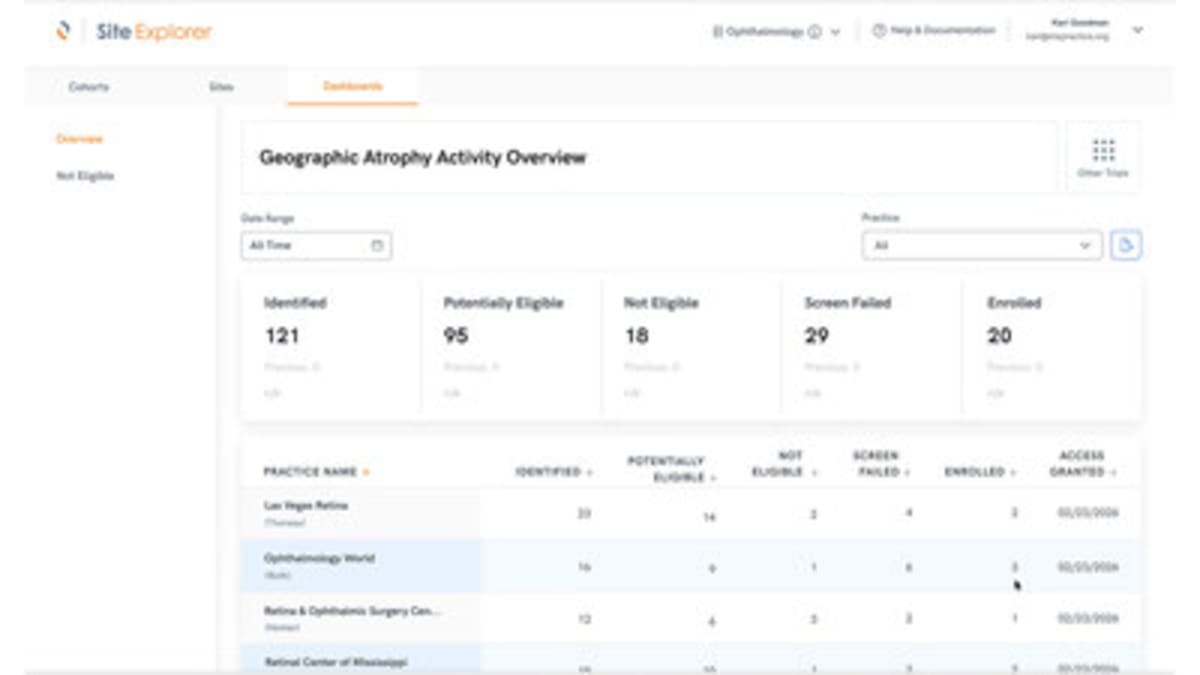
Verana Health announced new features in its Site Explorer and Verana Trial Connect platforms that give drug developers instant access to anonymized health records from 95 million patients. Instead of researchers manually combing through files at individual clinics, they can now see which medical practices have patients who match their study requirements.
The traditional approach has been painfully slow. Doctors and staff spend hours reviewing patient charts one by one to find people who qualify for trials. This manual process leads to missed deadlines and stalled research, sometimes adding years before new treatments become available.
The new system changes that entirely. It automatically checks patient records against study requirements and flags eligible candidates in real time. Researchers can also see how changing their criteria might expand or shrink their potential participant pool before committing to a trial design.
Dr. Michael Yen, a professor at Baylor College of Medicine, captured the impact simply: "Verana Trial Connect has totally changed the game. I no longer have to go through Epic to find patients."

The platform draws from exclusive partnerships with major medical registries in ophthalmology and urology, representing more than 20,000 doctors across 3,200 practices. The data refreshes regularly, giving researchers current information rather than outdated snapshots.
The Ripple Effect
When clinical trials move faster, patients gain access to potentially life-saving treatments sooner. A drug that might have taken eight years to reach approval could arrive in six. For someone with a progressive disease, those two years can mean everything.
The system also opens trial participation to more people. Patients whose doctors practice in smaller clinics now have the same chance to join studies as those near major research hospitals. This broader reach means trials can include more diverse participants, which leads to treatments that work better for everyone.
Verana Health CEO Sujay Jadhav noted that trials built on real-world data start stronger, avoiding the mismatched enrollment and unexpected complications that derail traditional studies. In an era when multiple companies compete for limited research sites, having accurate patient information upfront makes the difference between success and costly failure.
The technology is already being used in eye care and urology studies, with researchers reporting significant time savings in patient recruitment. As more medical specialties adopt similar approaches, the entire drug development pipeline could accelerate, bringing hope to millions waiting for better treatments.
More Images

Based on reporting by Google News - Clinical Trial Success
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it