Scientists Cloak Mitochondria to Treat Deadly Disease in Mice
Researchers discovered how to disguise healthy mitochondria so they can slip into diseased cells undetected, extending the lives of mice with a fatal genetic disorder. The breakthrough could one day help children born with rare mitochondrial diseases.
Scientists have figured out how to sneak healthy mitochondria into sick cells by wrapping them in a clever disguise, opening new possibilities for treating rare genetic diseases that currently have no cure.
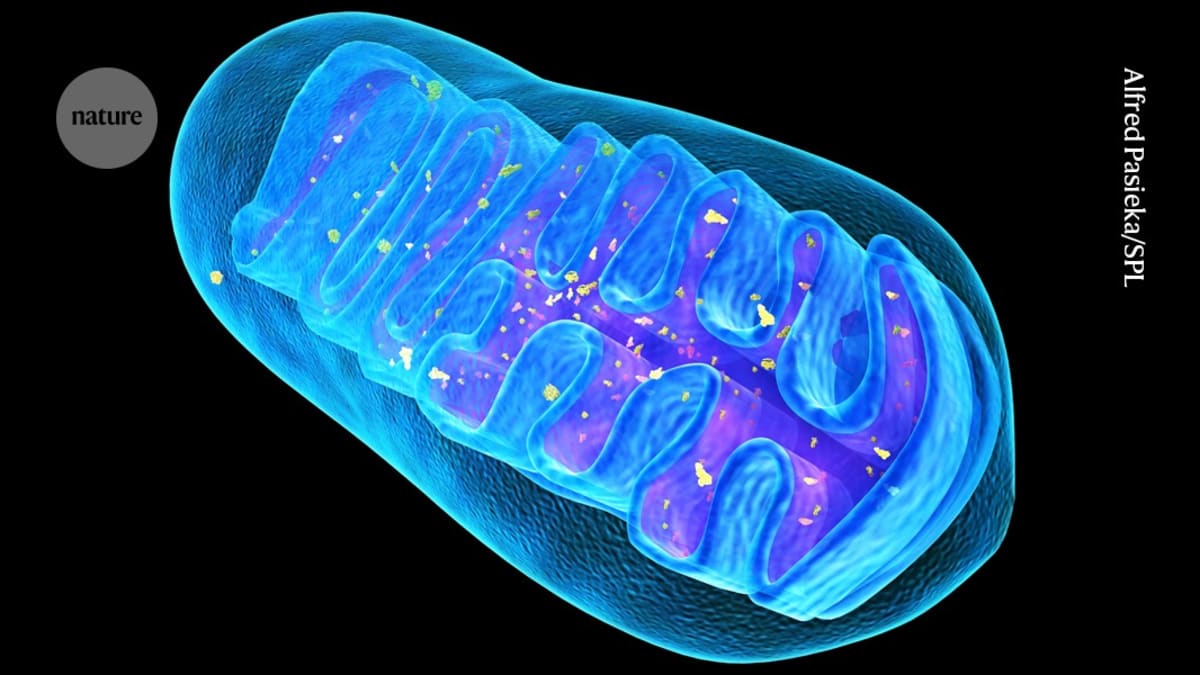
Mitochondria are the tiny powerhouses inside our cells that keep everything running. When they're damaged by genetic mutations, the results can be devastating, especially in diseases like Leigh syndrome, a rare disorder that often kills children in their early years.
For decades, researchers have tried transplanting healthy mitochondria into diseased cells, but the body's defense system kept destroying them. Cells recognize exposed mitochondria as damaged intruders and immediately tag them for destruction. Less than 5% of cells would accept the transplants using traditional methods.
The breakthrough came when researchers at Guangzhou Medical University wrapped mitochondria in membranes from red blood cells. The disguise was surprisingly simple to create: they just mixed ruptured red blood cells with isolated mitochondria. This protective shell preserved the mitochondria's electrical gradient, the signal that tells cells "I'm healthy and belong here."
The results were dramatic. About 80% of cells growing in lab dishes absorbed the cloaked mitochondria, compared to less than 5% with previous methods. Mike Devine, a neurobiologist at the Francis Crick Institute in London who wasn't involved in the study, called the difference "like night and day."

When tested in mice with a model of Leigh syndrome, the treatment extended their lives by about two weeks, roughly 20% longer than mice receiving unprotected mitochondria. While that might not sound like much, it's a significant step forward for a field that has struggled to make transplanted mitochondria work at all.
The Ripple Effect
This technique could transform how doctors approach dozens of rare mitochondrial diseases that affect roughly one in 5,000 people worldwide. Previous methods required such massive doses of mitochondria that scaling up to treat humans seemed nearly impossible. The new approach uses far smaller amounts while achieving much better results.
The method also opens doors beyond rare genetic diseases. Researchers are exploring whether mitochondrial health plays a role in more common conditions like Parkinson's disease, heart disease, and aging itself. If healthy mitochondria can be efficiently delivered to struggling cells, the applications could extend far beyond what scientists originally imagined.
Some experts urge caution about overstating the results, particularly claims about preventing Parkinson's in mice. But even skeptics acknowledge the advance is remarkable and addresses a fundamental problem that has blocked progress in mitochondrial medicine for years.
The path from mice to human patients is long and requires much more research, but scientists now have a tool that actually works at a practical level.
More Images
Based on reporting by Nature News
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


