China Approves World's First Brain Chip for Paralysis
For the first time ever, a brain implant has been approved for widespread use outside clinical trials, giving hope to thousands living with paralysis. China's approval of the NEO device marks a breakthrough moment for people with severe spinal cord injuries who previously had no treatment options.
Imagine being unable to move your arms or legs, then using just your thoughts to control a glove that lets you eat, drink, and regain independence.
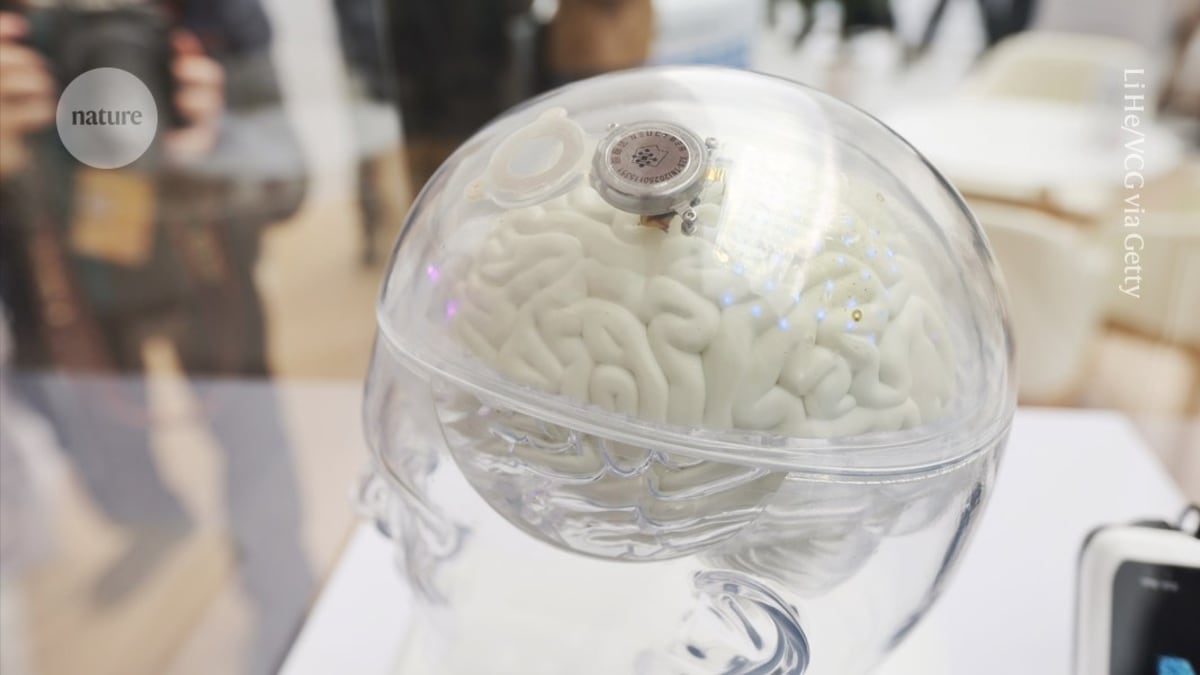
That future arrived last week when China approved NEO, a coin-sized brain implant developed by Shanghai-based Neuracle Medical Technology. It's the first brain-computer interface cleared for general use anywhere in the world, not just research trials.
The device works like a translator between brain and body. Eight tiny electrodes sit on the skull, reading electrical signals when someone imagines moving their hand. Those signals travel to a computer that decodes the thought and controls a soft robotic glove, letting paralyzed hands grasp objects again.
The approval covers people aged 18 to 60 with complete paralysis in all four limbs caused by neck-level spinal cord injuries. For these patients, effective treatments simply haven't existed until now, says neurosurgeon Chen Liang from Huashan Hospital, who helped run the trials.
In the clinical studies, all 32 participants gained the ability to perform grabbing movements they couldn't do before the implant. One person who used NEO for nine months regained enough hand function to eat and drink independently, tasks that had been impossible after their injury.

The device even sparked unexpected improvements. That same participant's left hand, which wasn't connected to the glove, also showed better movement, suggesting the brain may be rewiring itself in response to the technology.
The Ripple Effect
What sets NEO apart from competitors like Neuralink is its gentler approach. The device sits on top of the brain rather than being inserted into brain tissue, making surgery less risky and potentially speeding regulatory approval.
The research team has 18 months of data proving the system works long-term, rare evidence in a field where most devices remain experimental. That track record convinced regulators the technology was ready for real-world use.
Brain-computer interface researcher Zhengwu Liu calls the approval "a milestone for the whole field," noting it proves these devices can move from laboratory curiosity to medical reality. Several other BCIs are progressing through trials, including Paradromics' speech-restoration device and Neuralink's system, which enrolled 21 people in 2024.
BCI researcher Avinash Singh notes the study cohort remains small, but confirms the device appears both safe and effective. The technology offers something precious to people with paralysis: the chance to perform simple daily tasks most of us take for granted.
For thousands living with spinal cord injuries, the approval signals that thought-controlled movement isn't science fiction anymore.
More Images

Based on reporting by Nature News
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


