Drug Trial Brings Hope to 200,000 Heart Patients
A pharmaceutical breakthrough just opened doors for patients with a heart condition that's never had an approved treatment. Cytokinetics' drug Myqorzo succeeded in treating non-obstructive hypertrophic cardiomyopathy, doubling the number of people who could benefit from this therapy.
After nearly three decades of research, a biotech company just achieved what no one else has: positive trial results for a heart condition affecting roughly 200,000 Americans with no approved treatment options.
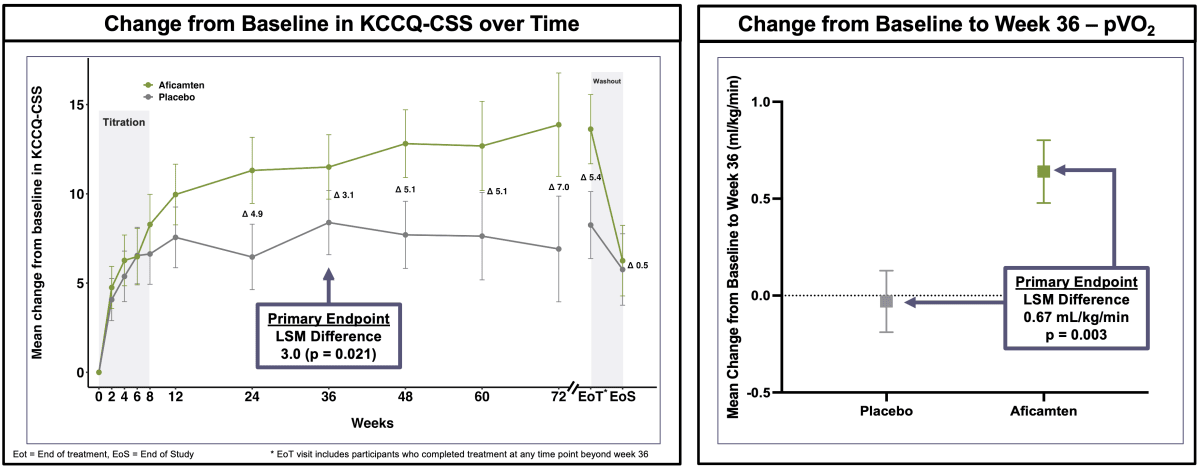
Cytokinetics announced Monday that its drug Myqorzo successfully met both primary goals in a phase 3 trial for non-obstructive hypertrophic cardiomyopathy (nHCM). This form of the heart disease makes the heart muscle abnormally thick, causing fatigue, shortness of breath, and reduced exercise capacity.
The trial measured two key improvements in patients taking Myqorzo compared to those on placebo. First, patients reported significantly better quality of life using a standard heart disease questionnaire. Second, their exercise capacity improved measurably through oxygen uptake tests.
Dr. Fady Malik, the company's research chief, described the exercise improvement as equivalent to "weeks of hard training" but delivered through medication. For patients who struggle with basic daily activities, that's transformative.

The company already received FDA approval for Myqorzo in December 2025 to treat the obstructive form of the same condition. Now, this new data could extend treatment to patients with the non-obstructive version, effectively doubling the number of people who might benefit.
The Ripple Effect
This success carries extra weight because a competitor's drug with the same mechanism failed in a similar trial last year. Bristol Myers Squibb's Camzyos, which treats the obstructive form and generated over $1 billion in sales last year, didn't achieve positive results in non-obstructive patients.
Cytokinetics CEO Robert Blum noted that trial steering committee members used words like "transformational" and "home run" when discussing the results. The company's stock jumped 21% on the news.
What makes this particularly meaningful is that researchers had no roadmap. No previous study had shown positive results in this patient population, so designing the trial meant venturing into uncharted territory.
For the estimated 200,000 Americans living with nHCM who've been managing symptoms without disease-specific medication, this represents the first real treatment option on the horizon.
Based on reporting by Google News - Clinical Trial Success
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it