
Epilepsy Drug Cuts Sleep Apnea Episodes by 50% in Trial
Millions who struggle with clunky CPAP machines finally have hope for a more comfortable solution. A repurposed epilepsy drug reduced sleep apnea breathing interruptions by up to 50% in a major European clinical trial.
Millions of people with sleep apnea may soon have a game-changing alternative to bulky breathing machines that keep them tethered at night.
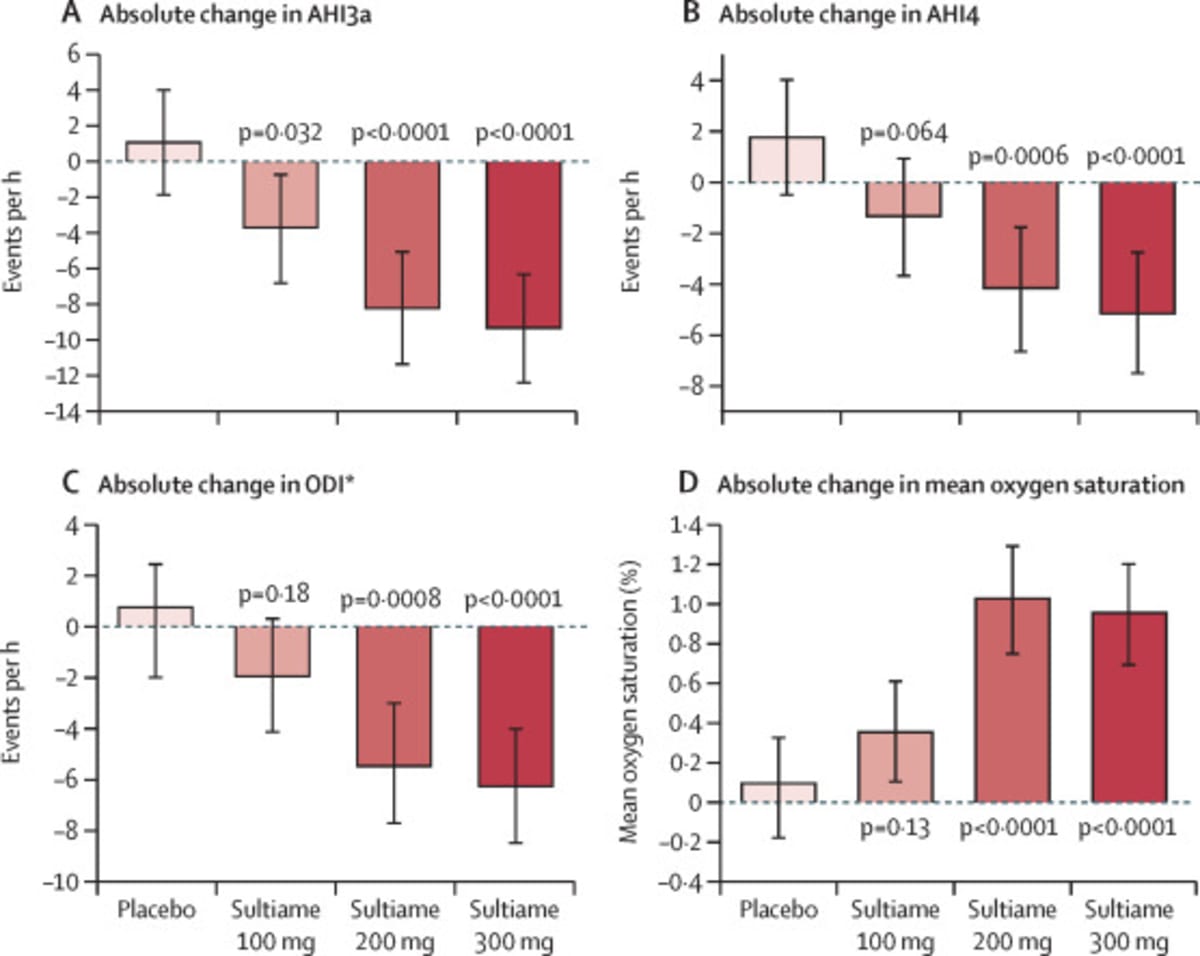
A large clinical trial across five European countries found that sultiame, a drug originally developed for epilepsy in the 1950s, dramatically reduced breathing interruptions during sleep. Participants taking the highest doses saw their sleep apnea severity drop by 30 to 50 percent.
The study involved 240 people with moderate to severe obstructive sleep apnea from Belgium, Czechia, France, Germany, and Spain. Researchers at the University of Gothenburg in Sweden divided participants into four groups, with three receiving different daily doses of sultiame (100 mg, 200 mg, or 300 mg) and one receiving a placebo.
Every group taking sultiame showed improvements in breathing disturbances, overnight oxygen levels, and daytime sleepiness compared to the placebo group. The sweet spot appears to be 200 mg daily, which delivers powerful results with minimal side effects.
"We have been working on this treatment strategy for a long time, and the results show that sleep apnea can indeed be influenced pharmacologically," says Jan Hedner, a pulmonary medicine specialist who led the research. "It feels like a breakthrough."

Unlike existing treatments that focus mainly on weight loss or mechanical devices, sultiame addresses root causes by improving muscle tone in the upper airways and stabilizing the body's respiratory control. This makes it the first drug to directly tackle the underlying mechanisms of obstructive sleep apnea.
Why This Inspires
Sleep apnea affects millions worldwide and increases risks for heart disease, cognitive problems, and diabetes. Current solutions like CPAP machines work well but many people abandon them because they're uncomfortable or inconvenient.
This breakthrough could make effective treatment cheaper, more accessible, and vastly more comfortable for people everywhere. While the FDA approved tirzepatide for sleep apnea in late 2024, that medication works primarily through weight loss rather than addressing airway function directly.
Researchers are now planning larger, longer-term studies to confirm sultiame's safety and effectiveness across broader patient groups. The drug is already approved for epilepsy treatment in Europe, Australia, Japan, and Israel, though not yet in the United States.
For the millions who've struggled to get restful sleep or couldn't tolerate existing treatments, a simple pill might finally offer the relief they've been dreaming of.
More Images


Based on reporting by Google News - Health
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


