FDA Fast-Tracks Sleep Apnea Patch That Maps Your Airway
A small facial patch that uses "sleep sonar" to pinpoint exactly where and why airways collapse during sleep just earned FDA breakthrough status. The AI-powered device could help 39 million Americans with sleep apnea get treatments tailored to their specific obstruction patterns.
Millions of people with sleep apnea struggle to find relief because current treatments take a one-size-fits-all approach to a highly individual problem. Now, a small patch worn on the face during sleep promises to change that by mapping exactly where each person's airway collapses.
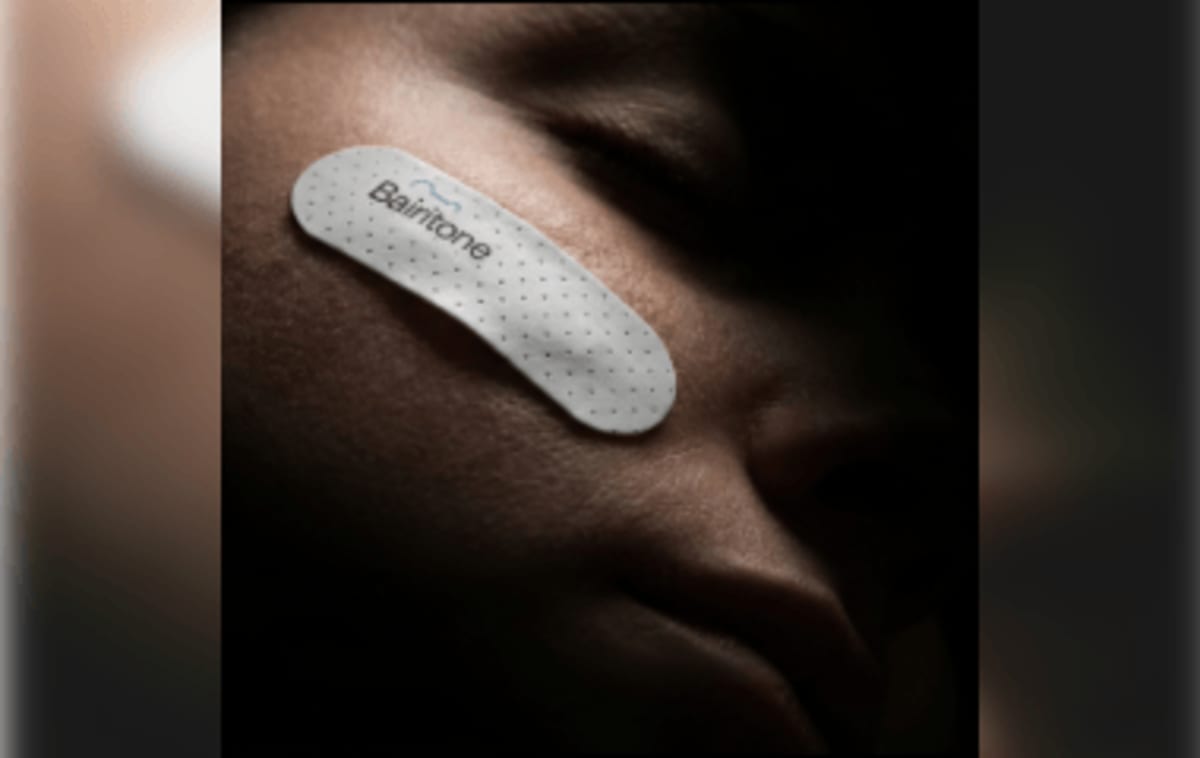
The FDA just granted Breakthrough Device Designation to Somnar, a sensor patch developed by Bairitone Health that captures the body's natural vibrations during sleep. Using what the company calls "sleep sonar," the patch detects subtle tissue movements with each breath and translates them into a dynamic map of upper airway behavior throughout the night.
Unlike traditional sleep studies that show breathing stops, Somnar reveals the anatomical why behind those pauses. The AI-powered platform tracks airway behavior across different sleep stages and body positions, identifying the specific sites and mechanisms causing obstruction in each individual.
The technology requires no MRI, X-ray, or ultrasound. Instead, it works like a highly sensitive microphone pressed against the skin, listening to vibrations that reveal how throat tissues behave during sleep. Bairitone Health calls it the first "anatomic polysomnogram."

For the 39 million Americans living with obstructive sleep apnea, this precision matters enormously. The condition causes airways to collapse repeatedly during sleep, leading to oxygen deprivation, disrupted rest, and increased risks of heart disease, stroke, and diabetes. Current treatments often fail because they don't account for individual anatomical differences.
The Breakthrough Device Designation means the FDA will prioritize Somnar's development and review process, potentially bringing it to patients faster while maintaining rigorous safety standards. The program is reserved for devices addressing serious conditions with the potential to offer more effective diagnosis or treatment than existing options.
Somnar is currently in research and development and not yet cleared for clinical use. However, it's already being tested in investigational settings, and in November 2025, the platform won the diagnostics category in the inaugural AASM Sleep Disruptors Innovation Awards.
The Ripple Effect
Personalized sleep apnea treatment could transform more than just nighttime rest. Better sleep means sharper thinking, improved mood, and reduced healthcare costs from preventing downstream complications. When clinicians can see exactly what's causing each patient's obstruction, they can recommend targeted therapies instead of trial and error. That means fewer frustrated patients abandoning treatment and more people reclaiming healthy sleep.
This small patch represents a shift from treating sleep apnea as a generic condition to understanding it as a personal anatomical puzzle with solutions tailored to each piece.
More Images




Based on reporting by Google News - Health Breakthrough
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


