First Human Vaccine for Top Bacterial Diarrhea Passes Tests
After 20 years of work, University of Guelph scientists have successfully tested the world's first human vaccine against campylobacter jejuni, the number one bacterial cause of diarrhea worldwide. The breakthrough could protect travelers, children in developing nations, and millions who suffer from this preventable illness each year.
A vaccine that could end the misery of millions suffering from severe food poisoning just passed a major milestone.
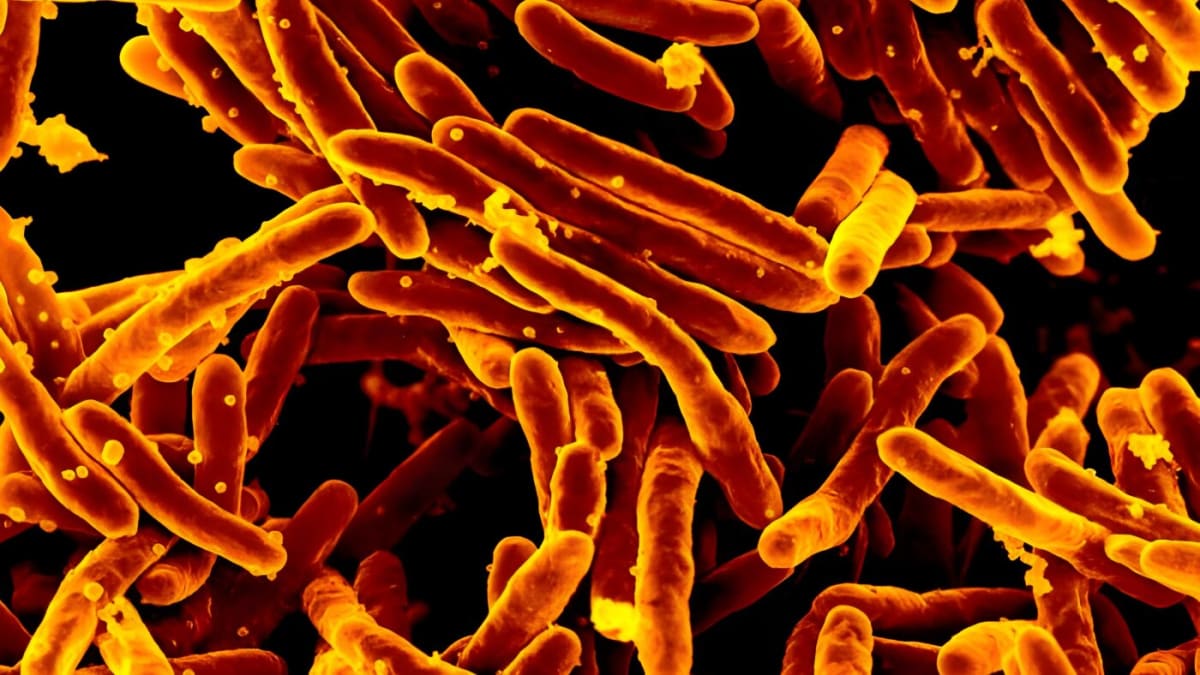
University of Guelph researchers have successfully completed phase one human trials for the world's first vaccine against campylobacter jejuni, the leading bacterial cause of diarrhea globally. The bacteria, commonly found in raw or undercooked chicken, causes severe cramps, fever, and diarrhea that can last for days and prove fatal if untreated.
"This campylobacter jejuni, it actually tops the list of the bacteria responsible for diarrhea illnesses in humans," said Mario Monteiro, professor in the department of chemistry who led the development. While salmonella and E. coli get more attention, campylobacter jejuni quietly infects more people worldwide.
The vaccine showed promising results when tested on about 60 healthy adults between 2022 and 2024 at Cincinnati Children's Hospital Medical Centre. Three doses successfully raised antibodies against the bacteria, with minimal side effects like muscle pain, tenderness, and fatigue.
The implications reach far beyond ruined vacations. In developing countries, campylobacter infections can stunt children's growth and prove especially dangerous for people with weakened immune systems. The World Health Organization has already recognized this vaccine as a preferred product.

What makes Monteiro's work unique is his sugar-based approach. His lab discovers sugar antigens and makes them immunogenic for use as vaccines, a technique that opens doors for creating multivalent products that could protect against multiple bacterial strains at once.
The 20-year journey from laboratory to human arms required patience and collaboration. Monteiro worked with Canadian researchers and the U.S. Naval Medical Research Center, with funding from the U.S. National Institutes of Health. This marks the fourth human trial for vaccines based on his discoveries.
The Ripple Effect
Between 30 and 40 graduate students contributed to this breakthrough over two decades. These young scientists learned that meaningful progress takes time, but their university lab work could protect people worldwide.
"At the university, we depend on graduate students," Monteiro said. "The beauty of this, too, is that it was all made in a university lab. And I think the students are proud to be involved."
Phase two trials will determine if the immune response actually prevents diarrheal illness by testing the vaccine against a placebo with hundreds of volunteers. This challenge trial will intentionally expose participants to live bacteria to measure protection. Phase three, still years away, would involve vaccinating hundreds of thousands of people.
For now, Monteiro and his students are celebrating a milestone two decades in the making and keeping their eyes on the next step forward.
Based on reporting by Google News - Vaccine Success
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it