Harvard Team Finds Hidden Switch in Cancer Drug Protein
Scientists discovered a secret "control panel" on a protein that powers billion-dollar cancer treatments, opening the door to safer, more targeted therapies. The breakthrough could let doctors destroy disease while protecting healthy cells.
A protein that's been fighting cancer for decades just revealed it has a hidden superpower.
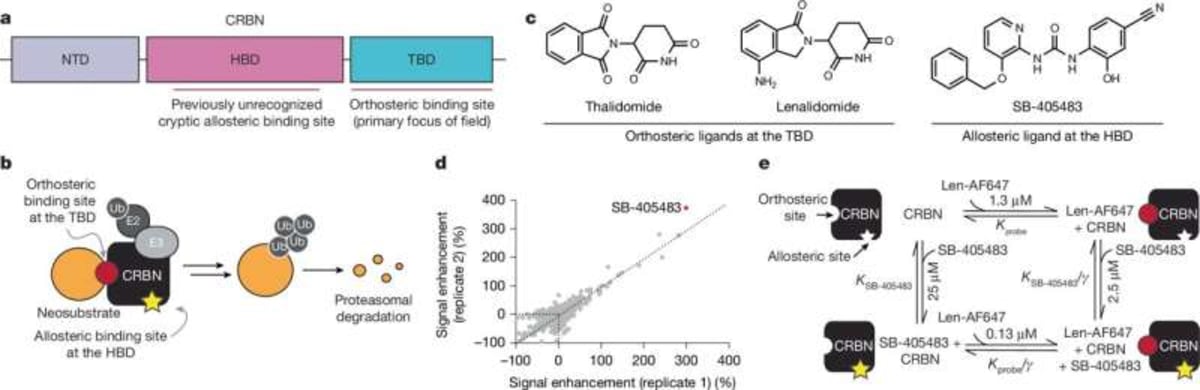
Researchers at Harvard University, working with teams at GSK and Scripps Research Institute, found a second binding site on cereblon, a protein that's already the engine behind multiple FDA-approved cancer drugs. Think of it like discovering your car has a second steering wheel that changes how the first one works.
Professor Christina Woo and her team published their findings in Nature this month. "When I share this work with audiences, you can see a ripple effect across the room of just how excited they are," Woo said.
Cereblon has a complicated history. It was accidentally targeted by thalidomide, the morning sickness drug that caused birth defects in the 1950s and 60s. But scientists later figured out how to harness that same targeting ability to treat blood cancers by directing cereblon to destroy disease-causing proteins.
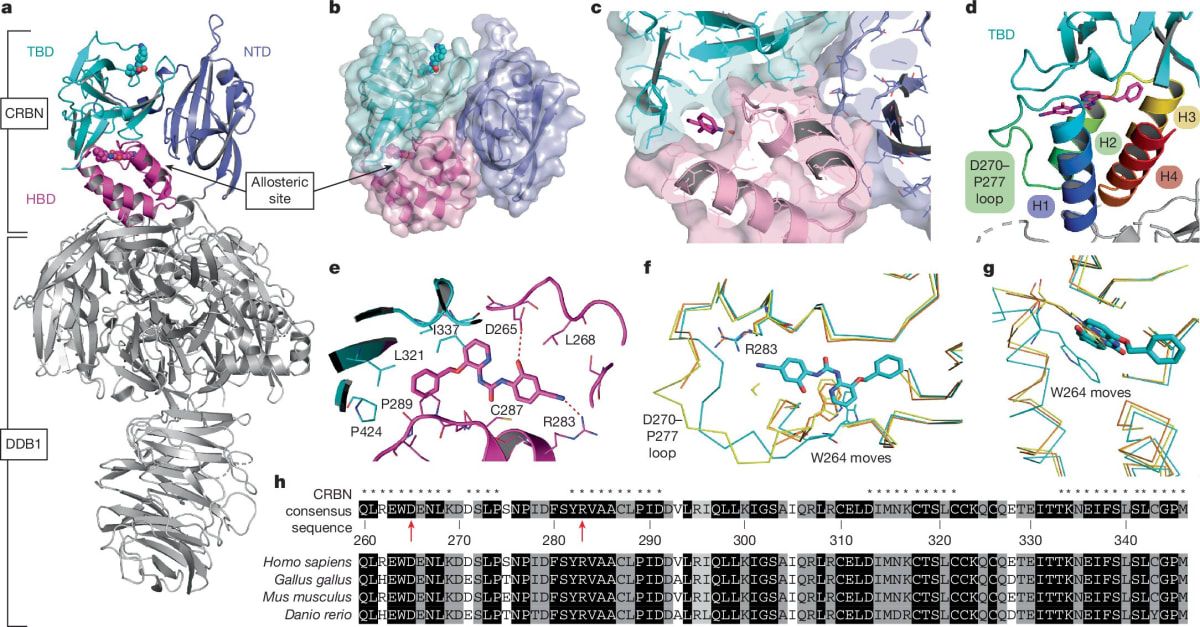
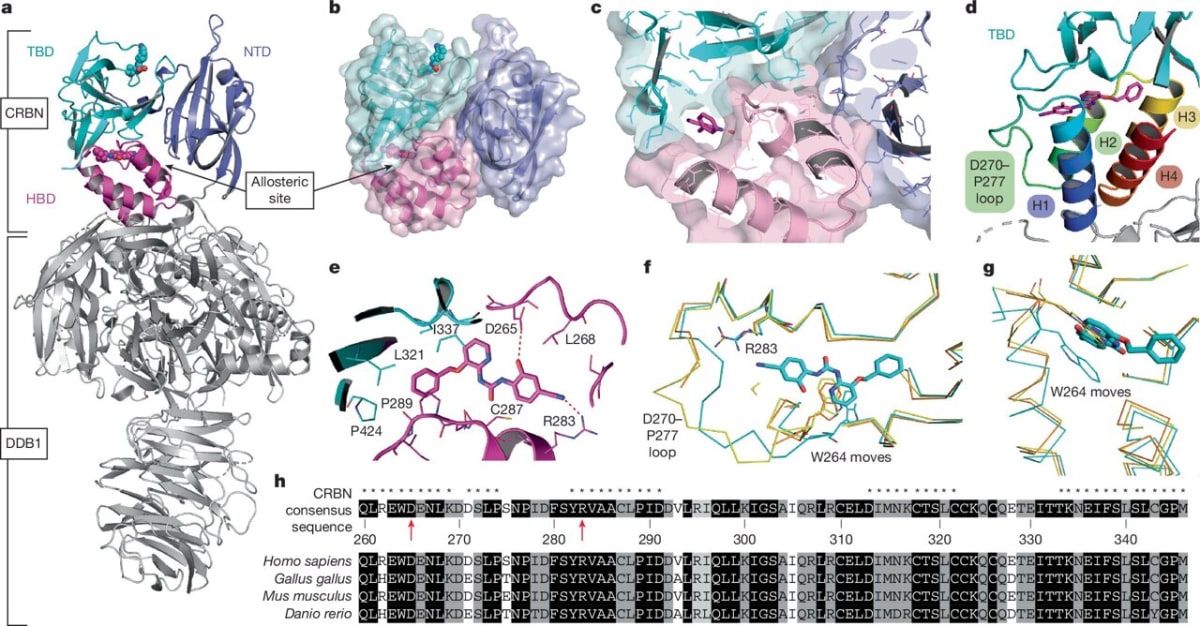
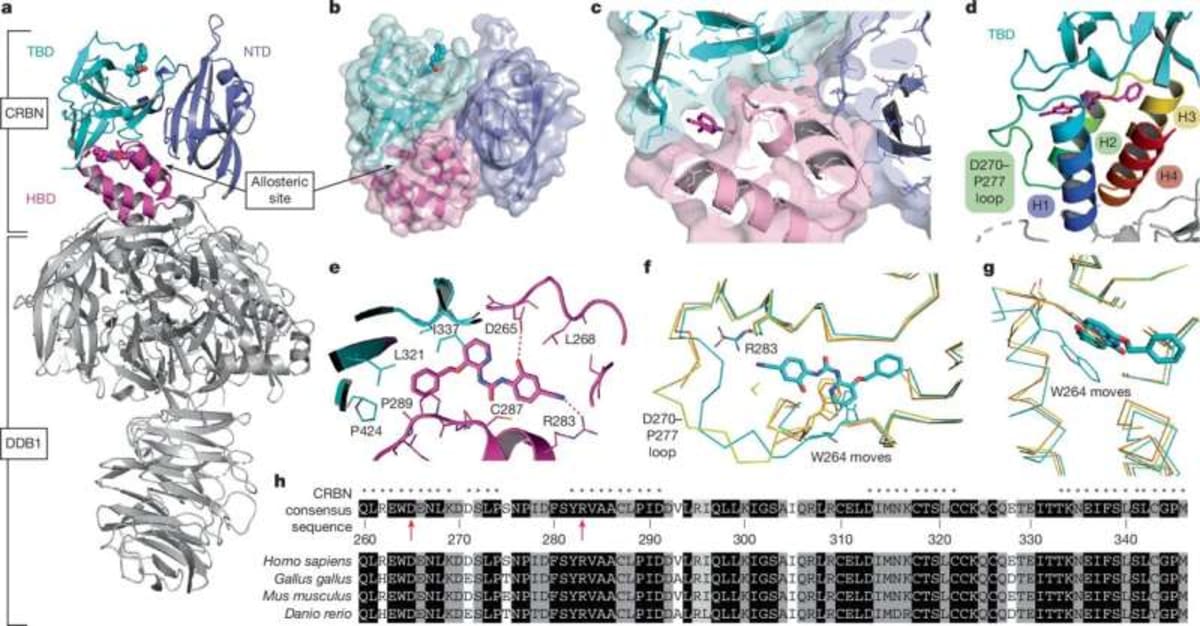
Until now, every drug designer focused on cereblon's main docking site. The new discovery shows there's a second pocket that acts like a dial, turning protein destruction up or down depending on what the cell needs.

Graduate student Vanessa Dippon led experiments using cells with fluorescent tags that glow when proteins are present and fade when they're destroyed. By adding different combinations of drugs to the main site and the newly discovered allosteric site, her team watched the glow patterns change in real time.
Sometimes the second site made cancer-causing proteins easier to destroy. Other times it protected healthy proteins from being broken down. The effect depended entirely on which drug combination they used.
The Ripple Effect
This precision matters because current cereblon drugs sometimes destroy proteins the body needs, causing side effects. The new binding site offers a way to boost a drug's cancer-fighting power while dialing down damage to healthy tissue.
The team used cryo-electron microscopy to capture high-resolution snapshots of cereblon's shape. When both binding sites are occupied, the protein shifts into a previously unknown form that's especially good at grabbing its targets.
The discovery opens two immediate paths forward. Doctors could add allosteric drugs to existing cancer treatments, making them work better with fewer side effects. Or drug designers could create entirely new molecules that use both sites at once, like molecular glue with two connection points instead of one.
For patients battling blood cancers and other diseases, this hidden pocket could mean the difference between a treatment that works and one that works well.
The future of precision medicine just got a new tool, and it was hiding in plain sight all along.
More Images

Based on reporting by Phys.org
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it