Huntington's Gene Therapy Shows 75% Slower Disease Progress
A groundbreaking gene therapy called AMT-130 has slowed Huntington's disease progression by 75% in clinical trials, offering hope to families who've waited 154 years for effective treatment. The one-time treatment could preserve patients' ability to walk, speak, and connect with loved ones for years longer.
For families watching loved ones lose the ability to speak, walk, and recognize them, a new gene therapy is delivering the hope they've desperately needed.
AMT-130, a one-time gene therapy treatment, slowed Huntington's disease progression by 75% in patients who received high doses during recent clinical trials. The results represent the first major breakthrough for a condition that has lacked disease-modifying treatments since it was first described in 1872.
Huntington's disease is a hereditary neurodegenerative condition that typically strikes people in their forties or fifties. Children of parents with Huntington's have a 50% chance of inheriting the disease themselves.
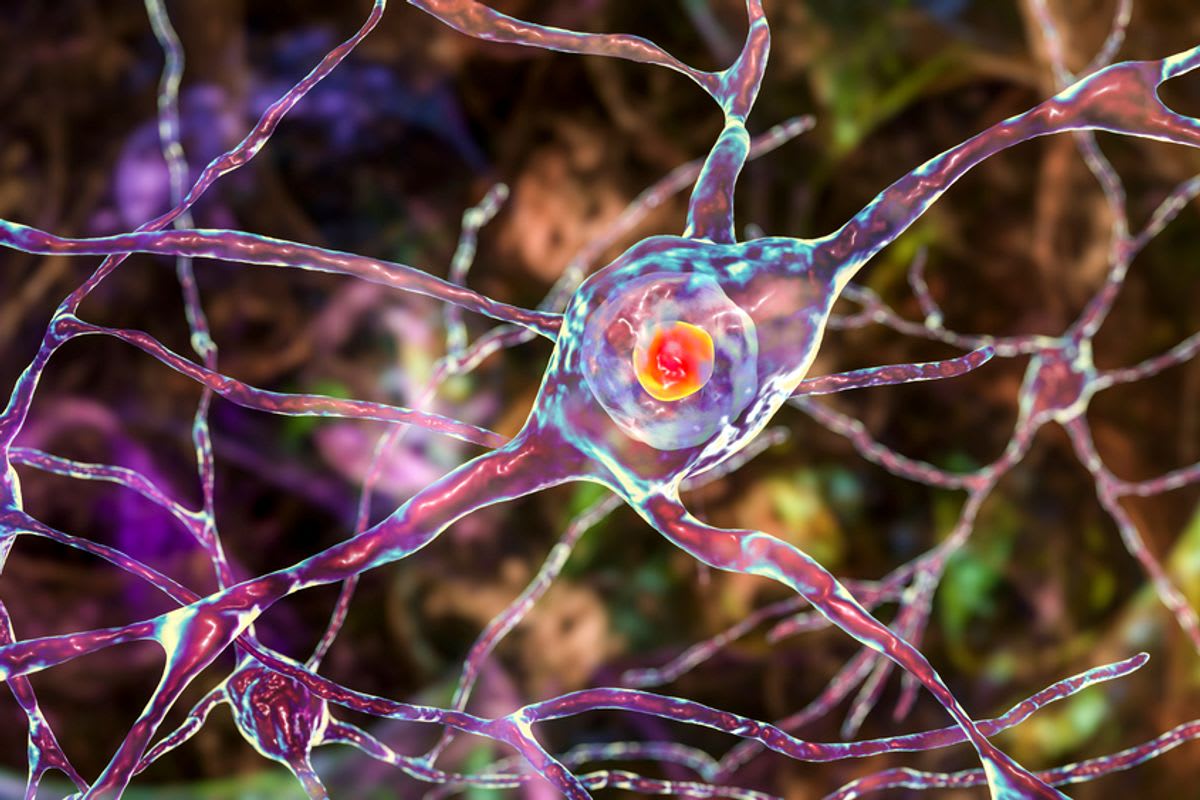
The condition causes involuntary movements, loss of speech and mobility, and psychiatric changes including irritability and aggression. Patients typically live 15 to 18 years after diagnosis, experiencing symptoms that combine the worst of Alzheimer's, Parkinson's, and ALS all at once.
The new treatment works by silencing the gene responsible for Huntington's. Administered through a single neurosurgical procedure, AMT-130 reduces the harmful protein that causes brain cell death and disease progression.

Seven patients at the University of Alabama-Birmingham received the high dose and reported better thinking, motor control, and ability to carry out daily functions three years later. Twelve study locations across the U.S., including Johns Hopkins and Rush University Medical Center, are now participating in trials.
"A therapy that can slow or stop progression of disease means preserving a person's ability to walk, speak, and connect with their loved ones, effectively giving families their futures back," said Amy Gray, president and CEO of the Huntington's Disease Society of America.
The Ripple Effect
The breakthrough extends beyond individual patients. Because Huntington's is hereditary, every month gained represents more time for parents to spend with children, more memories created, and more quality years for entire families across generations.
Scientists sequenced the Huntington's gene more than 30 years ago, but this marks the first treatment to actually slow the disease rather than just manage symptoms. The treatment hasn't yet been approved by the FDA, but advocates are hopeful the application currently in progress will bring relief to thousands of families soon.
After 154 years of watching Huntington's steal their loved ones, families finally have reason to believe the future could be different.
More Images

Based on reporting by Google News - Disease Cure
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it