
Lab-Grown Oesophagus Helps Pigs Swallow Again
Scientists successfully grew replacement oesophagi in the lab and transplanted them into pigs, restoring their ability to swallow and eat normally. The breakthrough could one day help children born with oesophageal defects avoid major surgery.
Imagine being able to grow a replacement organ from a patient's own cells, eliminating the need for invasive surgery or long transplant waiting lists. Scientists just made that vision a major step closer to reality.
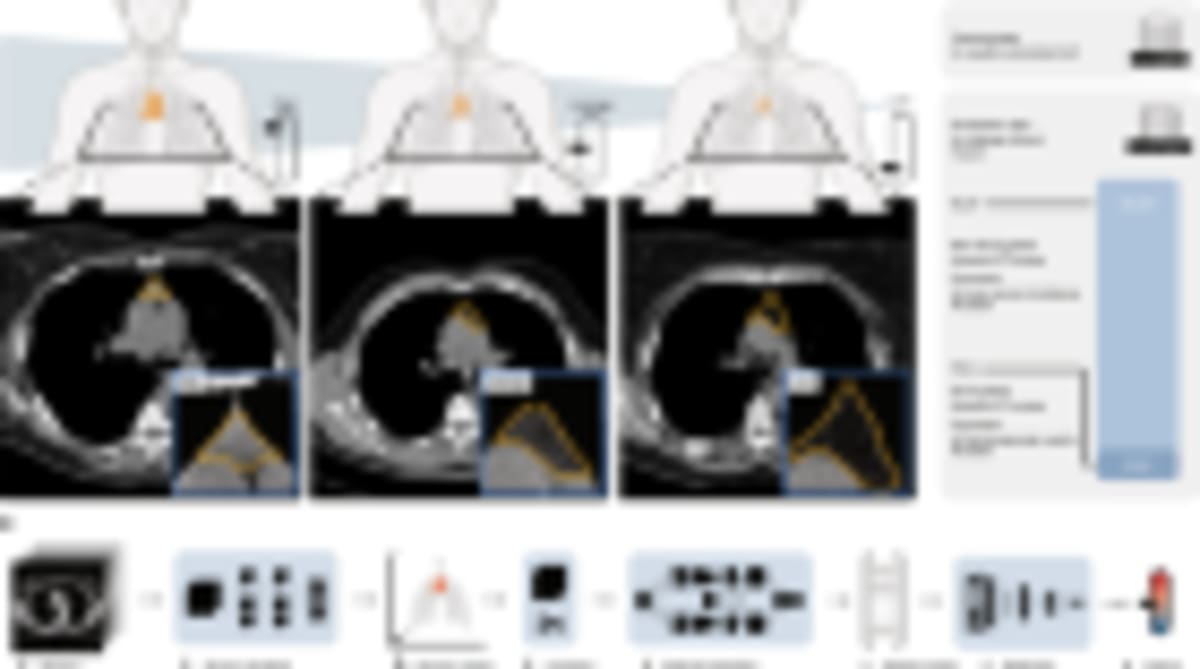
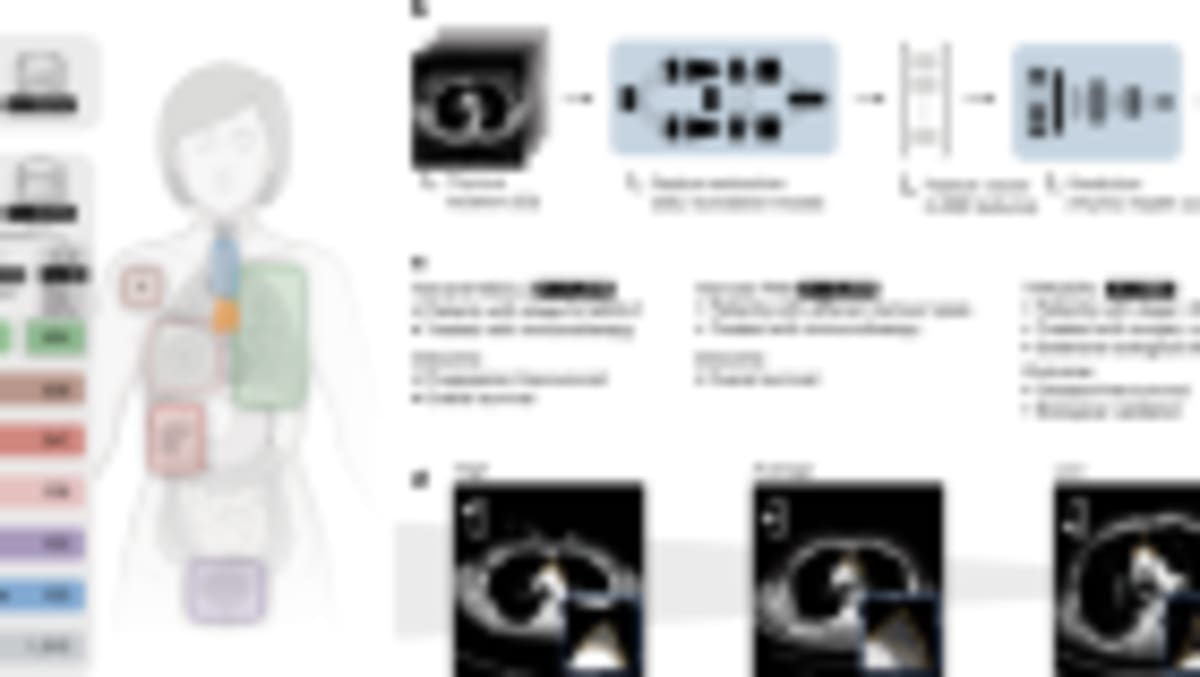
Researchers at University College London successfully created lab-grown oesophagi and implanted them into pigs, who went on to swallow and eat normally for six months. The team used stem cells from the recipient pigs to build the organs on natural scaffolds, creating grafts that developed functioning muscle, nerves, and blood vessels.
The work could transform treatment for children born with long-gap oesophageal atresia, a condition where a large section of the tube connecting the throat to the stomach is missing. Right now, surgeons have to move the child's stomach up to their neck or transplant part of their colon to bridge the gap. Both procedures are highly invasive and carry significant risks.
Lead researcher Paolo De Coppi and his team started with small samples of muscle cells and connective tissue from each pig. They transformed these into stem cells, then injected them into oesophagus scaffolds taken from other pigs. Over two months, the cells grew across the scaffolds to create complete grafts ready for transplant.

The surgeons replaced 2.5-centimeter segments of oesophagus in eight minipigs, chosen because their size approximates that of children who might one day receive this treatment. Five pigs survived the full six-month study period and showed normal swallowing function.
The Ripple Effect
This breakthrough builds on years of gradual progress. The team previously grew mouse cells on rat scaffolds and tested pig-based scaffolds in rabbits, but pigs offered the first realistic model for human physiology and size.
Andrew Barbour, an academic surgeon at the University of Queensland, called the ability to generate a fully functional oesophagus with all necessary components "impressive." He noted that while some scar tissue initially developed, it reduced over time, which suggests the technique could improve even further.
The research represents more than just one organ. The techniques developed here could potentially be applied to other tubular organs or tissues, opening doors for regenerative medicine across multiple conditions.
For families facing devastating diagnoses, this work offers something precious: hope that less invasive, more effective treatments are on the horizon.
More Images
Based on reporting by Nature News
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


