Scientists Create Stable Copper That Turns CO2 Into Fuel
Chinese researchers built an ultra-stable copper superatom that converts carbon dioxide into ethylene fuel with over 80% efficiency. This breakthrough could finally make industrial carbon recycling practical and affordable.
After years of setbacks, scientists have created something that could change how the world deals with carbon pollution: a tiny cluster of copper atoms that turns greenhouse gases into useful fuel without falling apart.
The challenge with copper has always been simple yet frustrating. It's cheap, abundant, and naturally good at binding carbon atoms together, making it perfect for converting carbon dioxide into industrial chemicals. But regular copper corrodes or breaks down almost immediately when exposed to air or the harsh conditions inside factories.
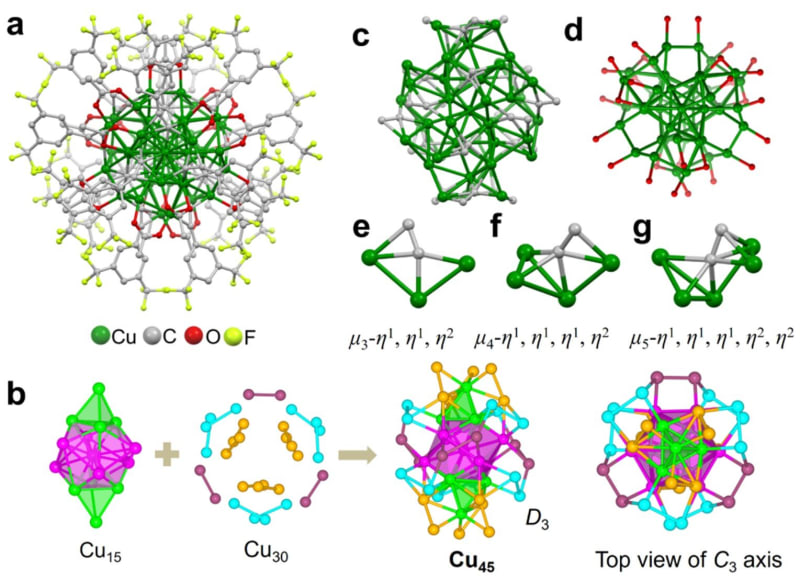
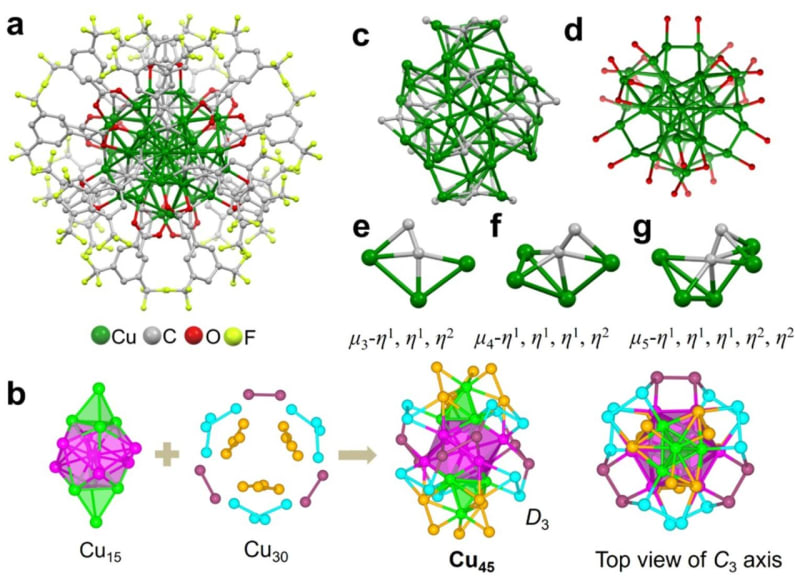
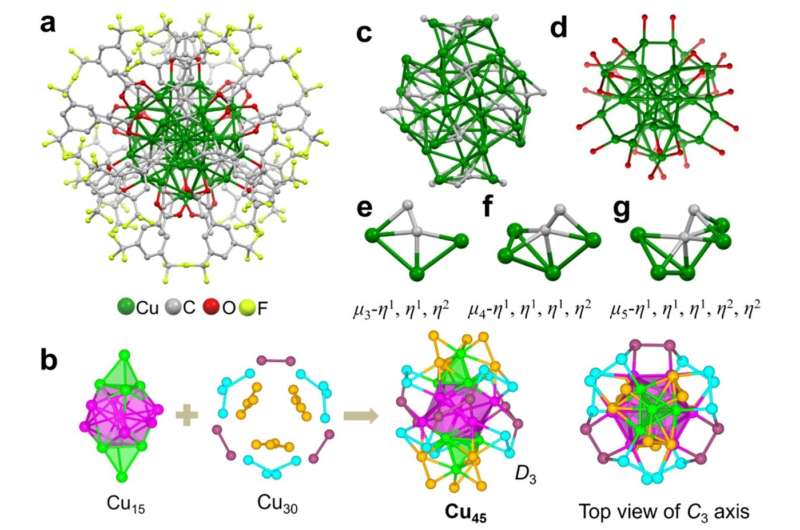
Researchers at Tsinghua University in Beijing solved this problem by building a superatom. They arranged 45 copper atoms into a special shell-like formation that mimics noble gases like neon or argon, which naturally resist reacting with their surroundings. Then they wrapped the entire cluster in protective organic molecules to keep it intact.
The team put their creation through brutal tests that would destroy ordinary copper. They submerged it in boiling water, dunked it in concentrated acid, exposed it to harsh chemicals, and heated it to 95°C. The copper cluster survived everything perfectly intact, showing toughness never before seen in a copper nanocluster.
Then came the real test. The researchers used electricity to break apart carbon dioxide molecules and let the copper superatom bind them back together in new forms. The result exceeded expectations: the cluster converted CO2 into ethylene with over 80% efficiency.

Ethylene matters because it's one of the world's most important industrial chemicals. Manufacturers use it to make everything from plastics to green fuels, producing over 200 million tons annually. Finding a cheap, stable way to create it from captured carbon dioxide instead of fossil fuels could reshape entire industries.
The Ripple Effect
This discovery goes beyond one impressive experiment. The research team has shown that creating stable copper catalysts is possible, opening doors for similar breakthroughs across chemistry and manufacturing.
Other research groups have spent decades trying to use copper for carbon recycling, only to watch their creations degrade too quickly for practical use. This stable superatom proves the concept can work outside the lab.
Industries that capture carbon dioxide from smokestacks or even directly from the air now have a potential pathway to turn that waste into valuable products. Instead of just storing captured CO2 underground, companies could transform it into materials the world needs.
The researchers believe their approach provides a blueprint for designing tough copper nanoclusters for many applications beyond carbon conversion. Each new stable catalyst brings us closer to a world where carbon emissions become raw materials instead of pollution.
One tiny cluster of 45 atoms might seem small, but sometimes the biggest changes start at the nanoscale.
More Images

Based on reporting by Google News - Science
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


