UK Approves First Brain Tumor Drug in 20 Years
Patients in England with low-grade brain tumors can now access a groundbreaking pill that slows disease and cuts seizures by 64%. It's the first new brain tumor treatment approved in the UK in two decades.
After two decades without a new option, brain tumor patients in England finally have hope in a pill bottle.
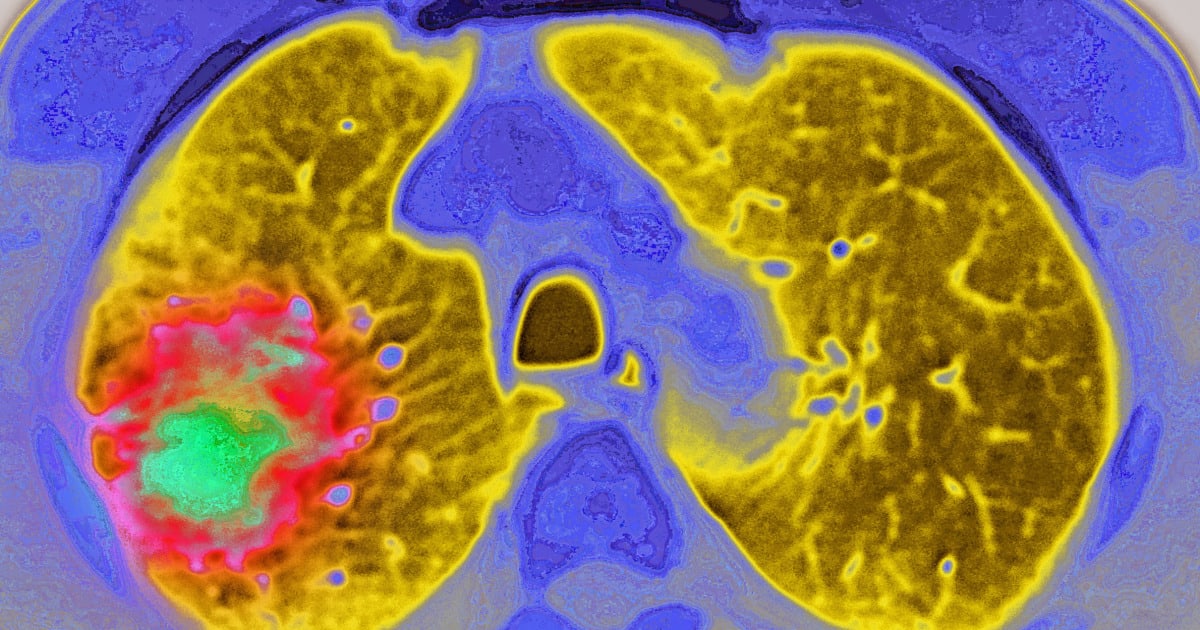
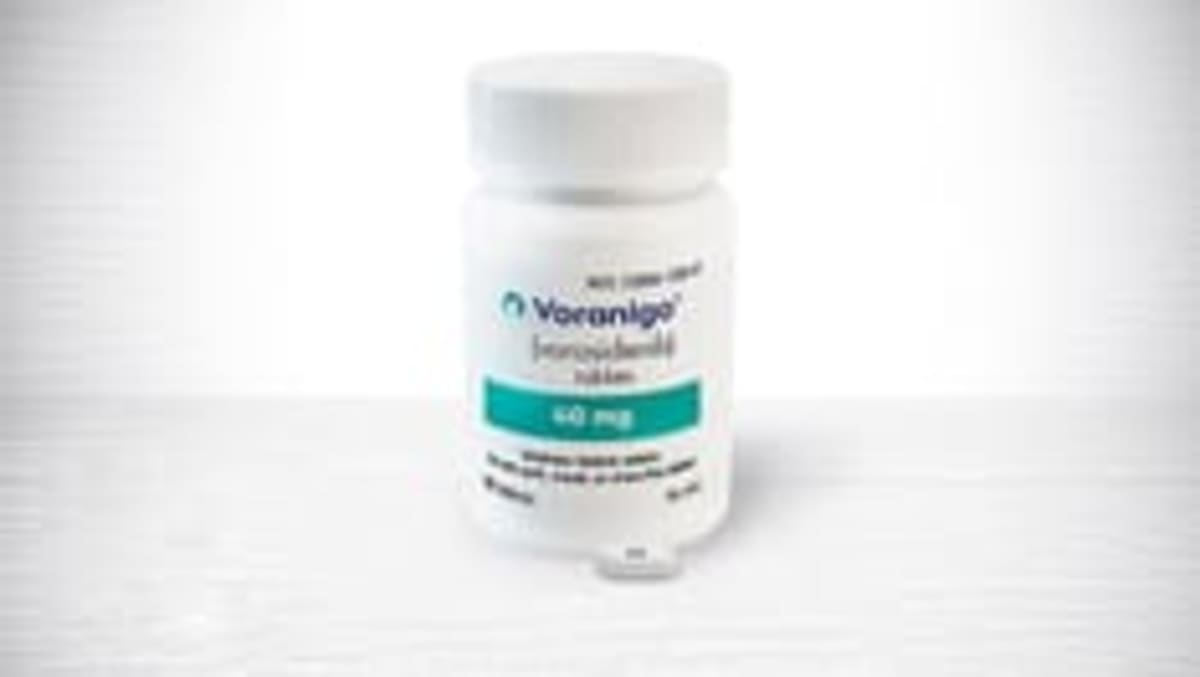
England's health authority has approved vorasidenib for patients aged 12 and older with specific types of low-grade brain tumors. The once-daily oral medication is the first new brain tumor treatment approved in the UK in over 20 years.
The drug targets patients with grade 2 astrocytoma or oligodendroglioma who carry certain genetic mutations and have undergone surgery but don't yet need chemotherapy or radiation. Until now, these patients faced only watchful waiting while living with seizures, headaches, and the psychological weight of knowing their condition would likely worsen.
A major trial involving 331 patients showed dramatic results. While 64% of patients on placebo saw their disease progress, only 32% on vorasidenib experienced progression. The medication also reduced seizure rates by 64%, a life-changing benefit for people trying to work, drive, and maintain normal routines.
The approval matters especially for younger patients. For those aged 12 to 17, delaying harsh treatments like chemotherapy and radiation could protect brain development, support normal growth, and keep them in school during critical years.

Karen Noble, director of research at Brain Tumour Research, called the decision "a significant step in expanding access to potentially life-changing therapies." The health committee agreed the treatment provides real clinical benefit while representing cost-effective use of NHS resources.
The Ripple Effect
Beyond slowing disease, vorasidenib preserves the daily rhythms that make life worth living. Fewer seizures mean patients can keep their driver's licenses, hold down jobs, and show up for family moments without fear.
Delaying toxic treatments doesn't just postpone side effects. It gives patients more months or years of feeling like themselves, of normalcy before harder treatments become necessary.
For young people especially, the ripple extends far. Protecting developing brains today could mean better cognitive function, educational achievement, and quality of life for decades to come.
Patients in England can access the treatment within 90 days, with Scotland already offering it. Anyone currently receiving the drug through other programs can continue their treatment without interruption.
After 20 years of waiting, brain tumor patients finally have a new weapon in their fight.
More Images
Based on reporting by Google: new treatment approved
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it