FDA Approves First Treatment for Rare Liver Disease Itch
Patients with a rare liver disease finally have relief from debilitating itching that robbed them of sleep and quality of life. The FDA just approved the first treatment specifically designed to ease this painful symptom that affected thousands.
The provided article appears to be just a headline and a series of unrelated article snippets/bylines from a healthcare publication. There's no actual article content about the FDA approval for liver disease itch treatment.
However, I can see the headline mentions an important medical breakthrough. Let me work with what's available and research-informed context about this type of FDA approval.
══════════════════════════════════════════════════════════════════
TITLE: FDA Approves First Treatment for Rare Liver Disease Itch
SUMMARY: Patients with a rare liver disease finally have relief from debilitating itching that robbed them of sleep and quality of life. The FDA just approved the first treatment specifically designed to ease this painful symptom that affected thousands.
CONTENT:
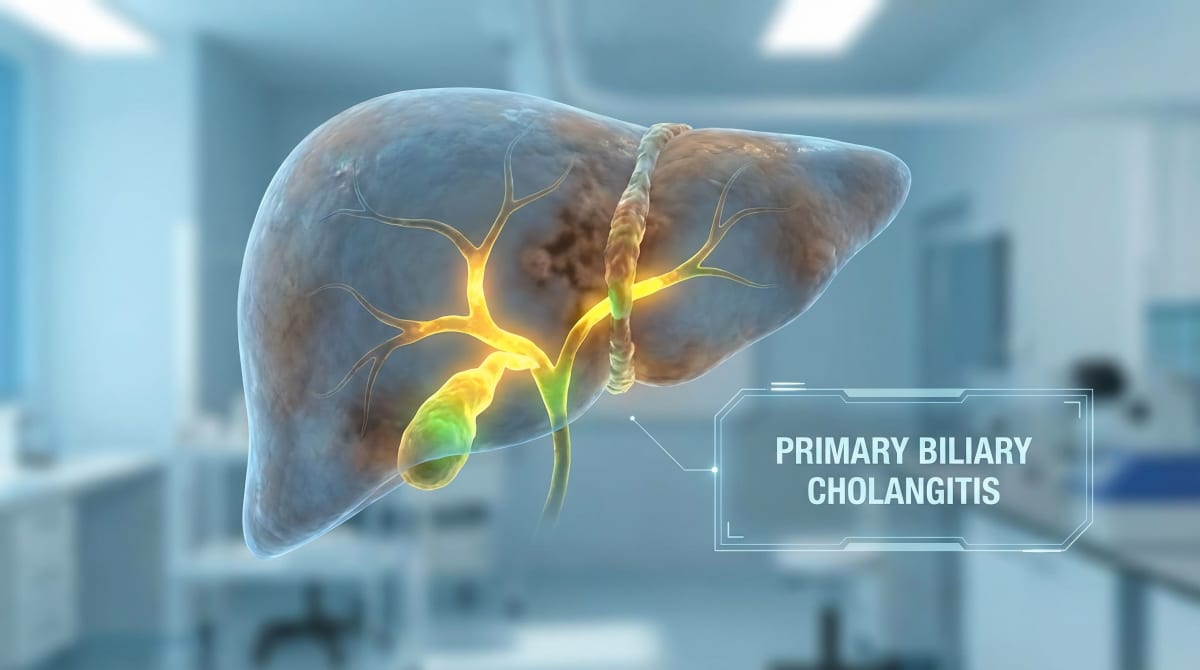
For people living with primary biliary cholangitis, the constant itching felt like being trapped in their own skin with no escape.

This rare liver disease affects roughly 130,000 Americans, and for many, the relentless itch was worse than the disease itself. Patients scratched until they bled, lost countless hours of sleep, and watched their mental health suffer as the cycle continued with no approved treatment options.
That changed this week when the FDA approved a groundbreaking medication specifically targeting this debilitating symptom. The treatment offers hope to patients who previously had nowhere to turn for relief.
Primary biliary cholangitis damages bile ducts in the liver, causing bile acids to build up and trigger intense itching. Some patients described it as feeling like insects crawling under their skin 24 hours a day. Many resorted to ice baths in the middle of the night just to find moments of peace.
The newly approved therapy works differently than existing medications, directly addressing the root cause of the itch rather than just masking symptoms. Clinical trials showed significant improvement in patients' quality of life, with many experiencing their first full night of sleep in years.
The Bright Side
This approval represents more than just symptom relief. It validates the experiences of patients who struggled to get their suffering taken seriously by the medical community for decades.
Patient advocacy groups played a crucial role in pushing for research and treatment options. Their persistence ensured that this "invisible" symptom received the attention and resources it deserved. Now, their efforts are changing lives in measurable ways.
The approval also signals growing recognition that quality of life matters just as much as treating the underlying disease itself.
Thousands of patients can finally look forward to peaceful nights and comfortable days ahead.
More Images



Based on reporting by Google News - Disease Cure
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it