Frozen Battery Breakthrough Could Store 50% More Energy
Scientists discovered that frozen electrolytes can power next-generation batteries, challenging everything we thought about how batteries work. This ice-like solution could make safer, more powerful lithium metal batteries a reality.
Researchers just turned conventional battery wisdom on its head by proving that frozen electrolytes can conduct electricity just as well as liquid ones.
A team from South Korea's UNIST and KAIST created what they call an "ice electrolyte" using ethylene carbonate, a common battery ingredient that's normally mixed with other liquids to keep it flowing. Instead, they kept it frozen solid at room temperature and discovered something remarkable: lithium ions could still move through it efficiently.
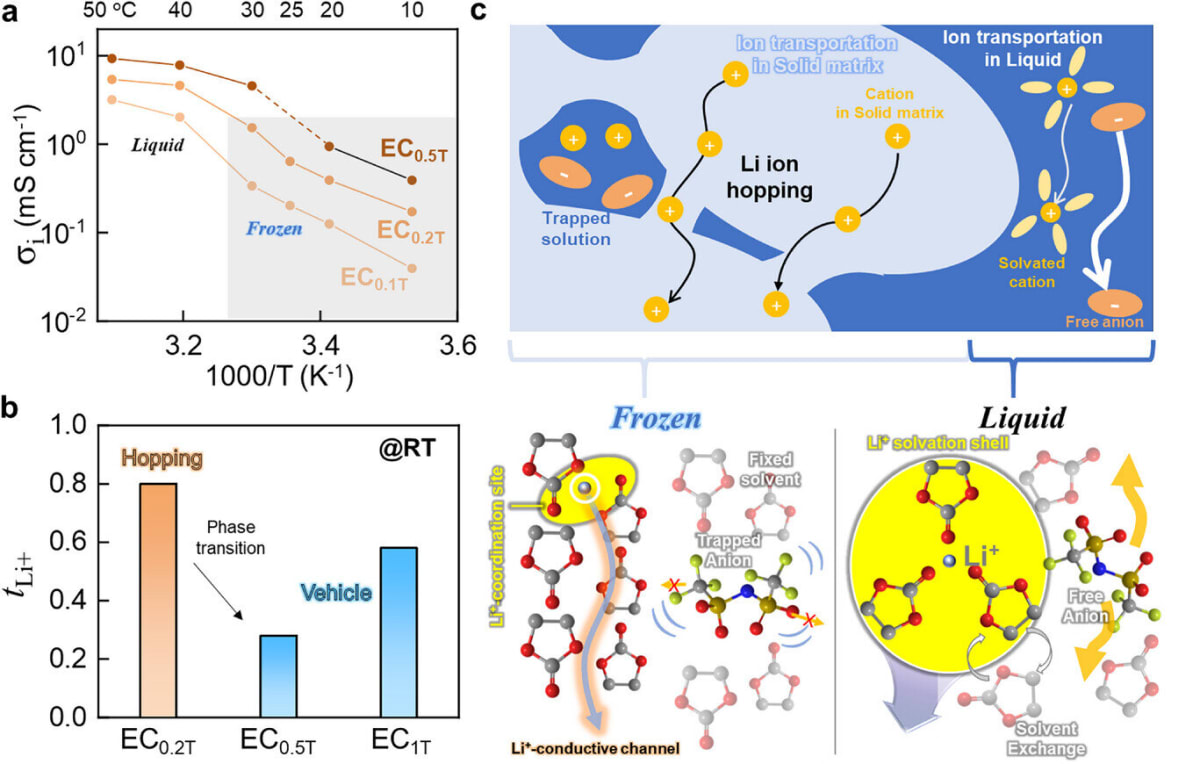
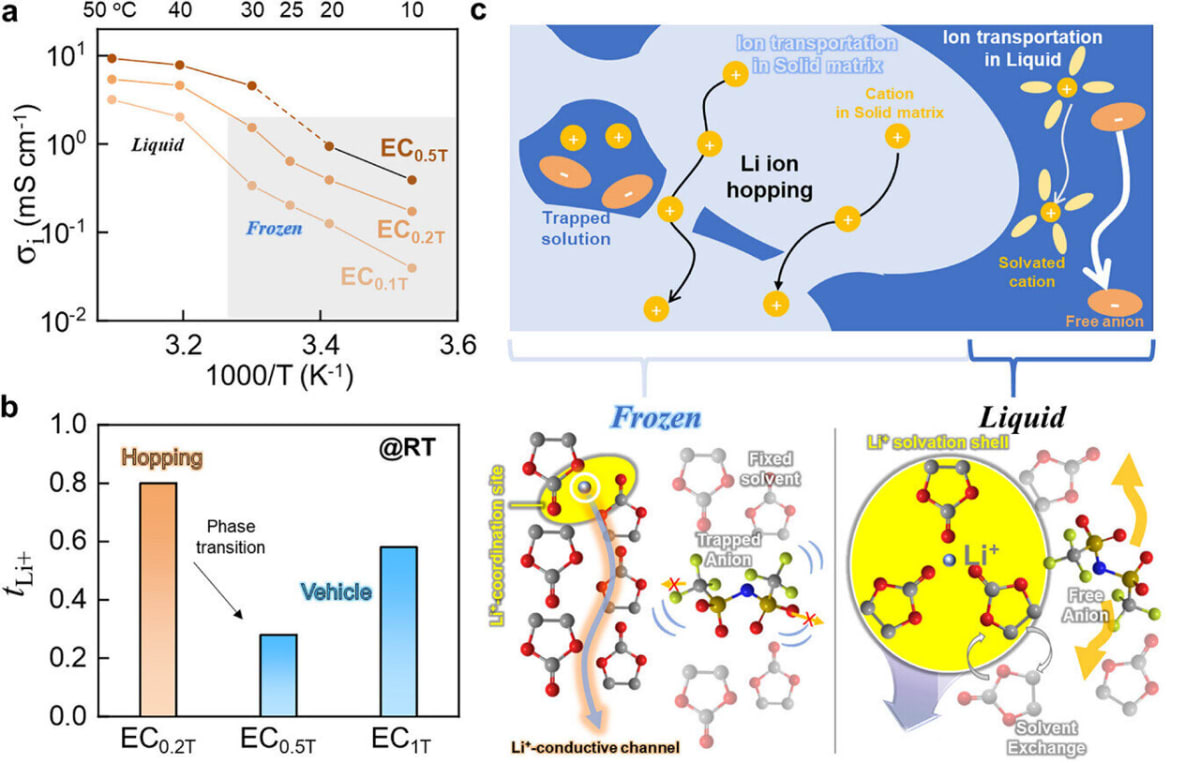
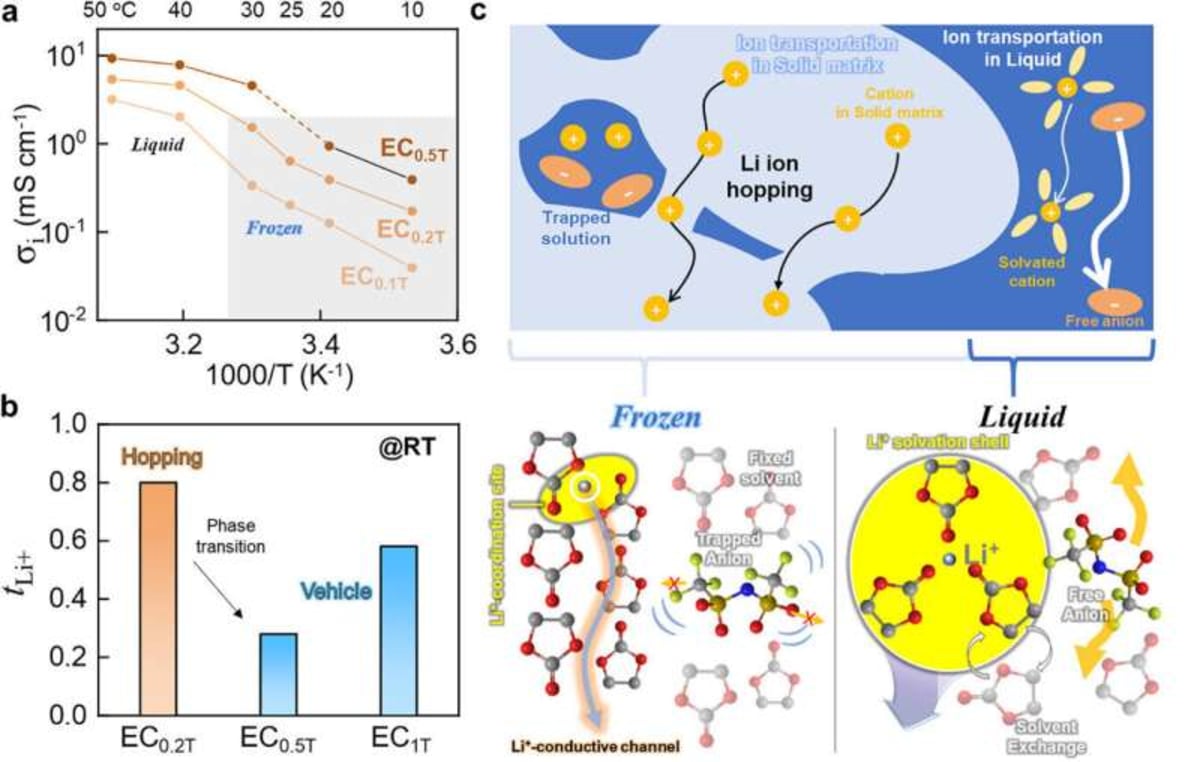
The frozen electrolyte uses a hopping mechanism where lithium ions jump between oxygen atoms in the immobilized solvent molecules. Think of it like stepping stones across a frozen pond, except the stones are perfectly arranged to guide ions exactly where they need to go.
The results speak for themselves. The ice electrolyte achieved an ionic conductivity of 0.64 mS/cm with a lithium-ion transference number around 0.8, matching the performance of advanced solid electrolytes. When tested in actual batteries, it powered over 400 charge and discharge cycles without failure.
This matters because lithium metal batteries represent the holy grail of energy storage, capable of holding up to 50% more power than today's lithium-ion batteries. But they've been plagued by dangerous problems: sharp lithium deposits called dendrites that puncture battery components and cause short circuits, plus harmful reactions between lithium metal and liquid electrolytes.

The Bright Side
The frozen structure solves both problems at once. The immobilized solvent molecules prevent dendrite formation by guiding ion movement in controlled pathways. No sharp deposits means no punctured separators and no battery failures.
Professor Hyun-Kon Song explains that solid electrolytes don't need to be rigid inorganic materials or special polymers. Even a loosely arranged, ice-like structure can support efficient ion conduction. His team is now exploring combinations of organic solvents with higher melting points to make this practical for everyday use.
The breakthrough opens doors for safer electric vehicles, longer-lasting smartphones, and more reliable grid-scale energy storage. While most researchers focused on developing complex polymer or ceramic electrolytes, this team found elegance in simplicity: just freeze what's already there.
The research was published in the journal Advanced Materials after peer review, confirming the scientific validity of this counterintuitive approach.
Sometimes the best solutions come from questioning our basic assumptions about how things should work.
More Images


Based on reporting by Phys.org - Technology
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


