Scientists Unlock Smarter Chemical Reactions With New Tool
Japanese researchers created a molecular tool that makes chemical reactions more precise and energy-efficient by turning weak forces on and off like a switch. This breakthrough could lead to better medicines and cleaner energy.
A team of scientists just solved a problem that's been holding back cleaner, more efficient chemistry for decades.
Researchers at Yokohama National University and the University of Tokyo designed molecules that don't just move electrons around. They actively control chemical reactions with the precision of a conductor leading an orchestra.
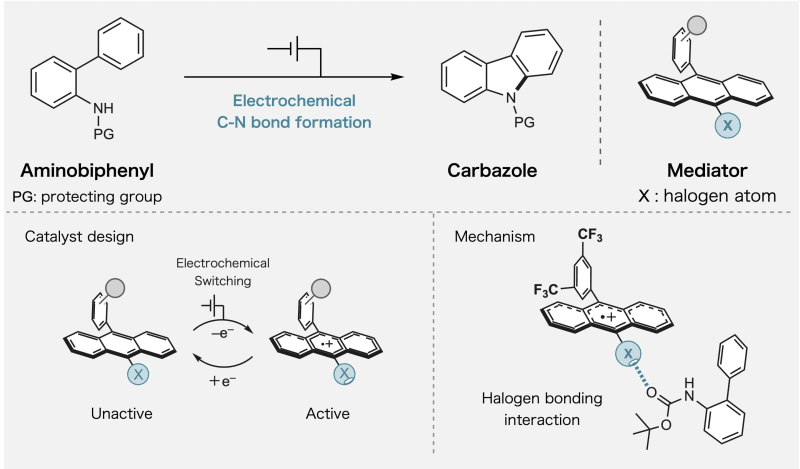
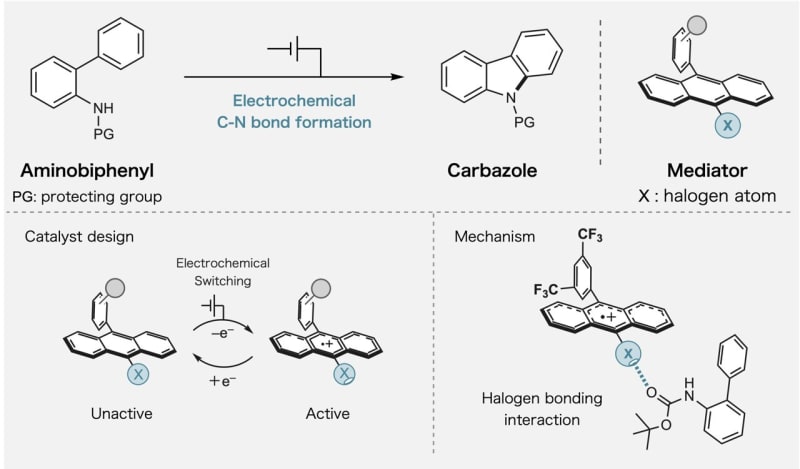
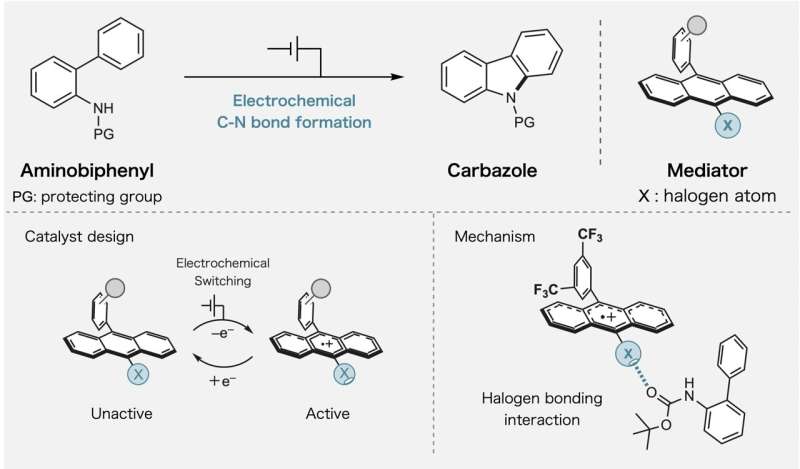
The breakthrough centers on something called halogen bonding, a weak force between molecules that the team learned to switch on and off. When they apply electricity to their specially designed molecules, these bonds activate and guide specific atoms to connect in exactly the right way.
Think of it like teaching molecules to be smarter helpers. Traditional molecules in these reactions act like simple delivery trucks, shuttling electrons from point A to point B. The new molecules act more like skilled architects, holding materials in place and making sure everything connects perfectly.
The team focused on creating bonds between carbon and nitrogen atoms, a process essential for making pharmaceuticals and advanced materials. Their method uses less energy and creates fewer unwanted byproducts than current techniques.
Professor Naoki Shida explained that most people thought redox mediators could only carry electrons. His team proved they can do much more by building control mechanisms directly into the molecules.

The researchers tested their approach using haloanthracene, a molecule where hydrogen atoms are swapped out for halogen atoms. When they added one electron to oxidize these molecules, the halogen bonding became much stronger, letting the molecules grab onto and organize other materials with remarkable precision.
Dr. Kayo Suda noted that while halogen bonding has been used in other areas of chemistry, nobody had explored its potential in redox catalysis until now. The combination turned out to be powerful.
Why This Inspires
This discovery represents a fundamental shift in how we approach chemical reactions. Instead of brute force methods that waste energy and create pollution, scientists now have a tool for elegant, targeted chemistry.
The implications stretch across industries. Pharmaceutical companies could design drugs with fewer side effects. Energy storage systems could become more efficient. Manufacturing processes could generate less waste.
Professor Mahito Atobe said the team's ultimate goal is creating a general platform that makes electrochemical synthesis more efficient, predictable, and sustainable. They're already planning to extend this concept to other types of bond-forming reactions.
The research, published January 8 in the Journal of the American Chemical Society, opens doors that scientists didn't even know existed. By proving that weak molecular forces can be switched on and off to control reactions, the team handed chemists everywhere a new design principle.
Clean energy technologies desperately need reactions that don't waste power. This breakthrough moves us closer to batteries, fuel cells, and chemical processes that work smarter instead of harder.
More Images


Based on reporting by Phys.org
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


