Virginia Doctors Test New Heart Device to Prevent Stroke
A Virginia heart center just became the first in the state to implant a foam device that could protect people with irregular heartbeats from strokes. The innovative treatment offers hope for patients who can't take blood thinners.
Doctors in Virginia are testing a breakthrough device that could save thousands of lives by preventing strokes in people with irregular heartbeats.
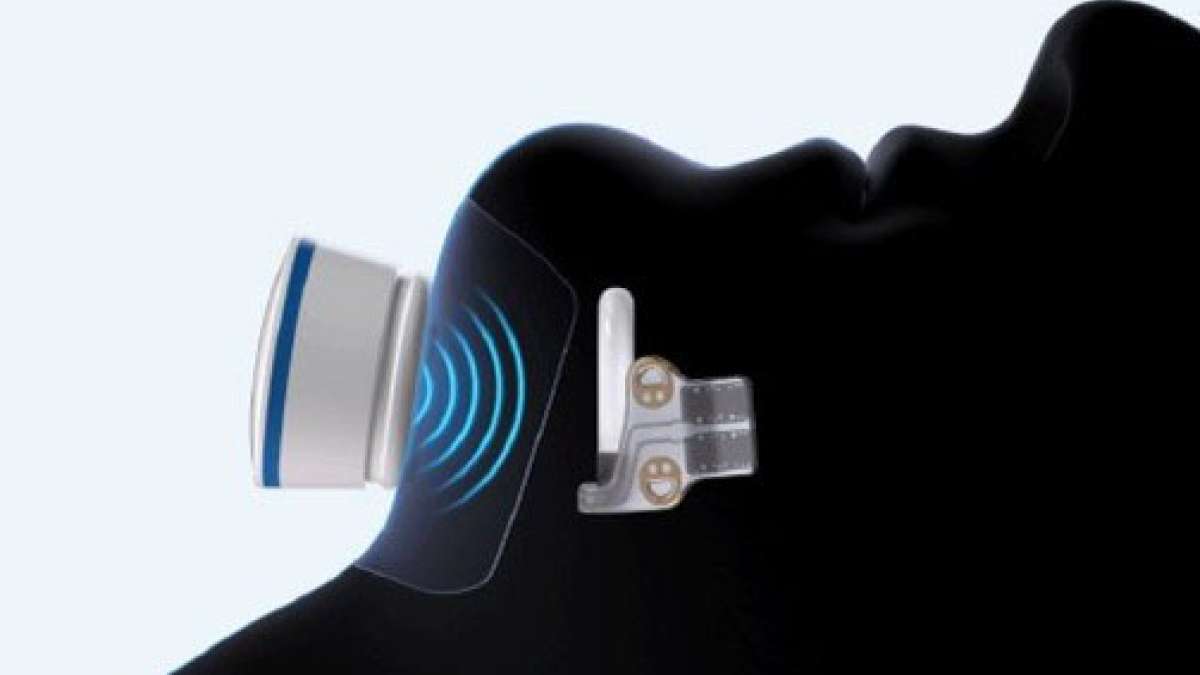
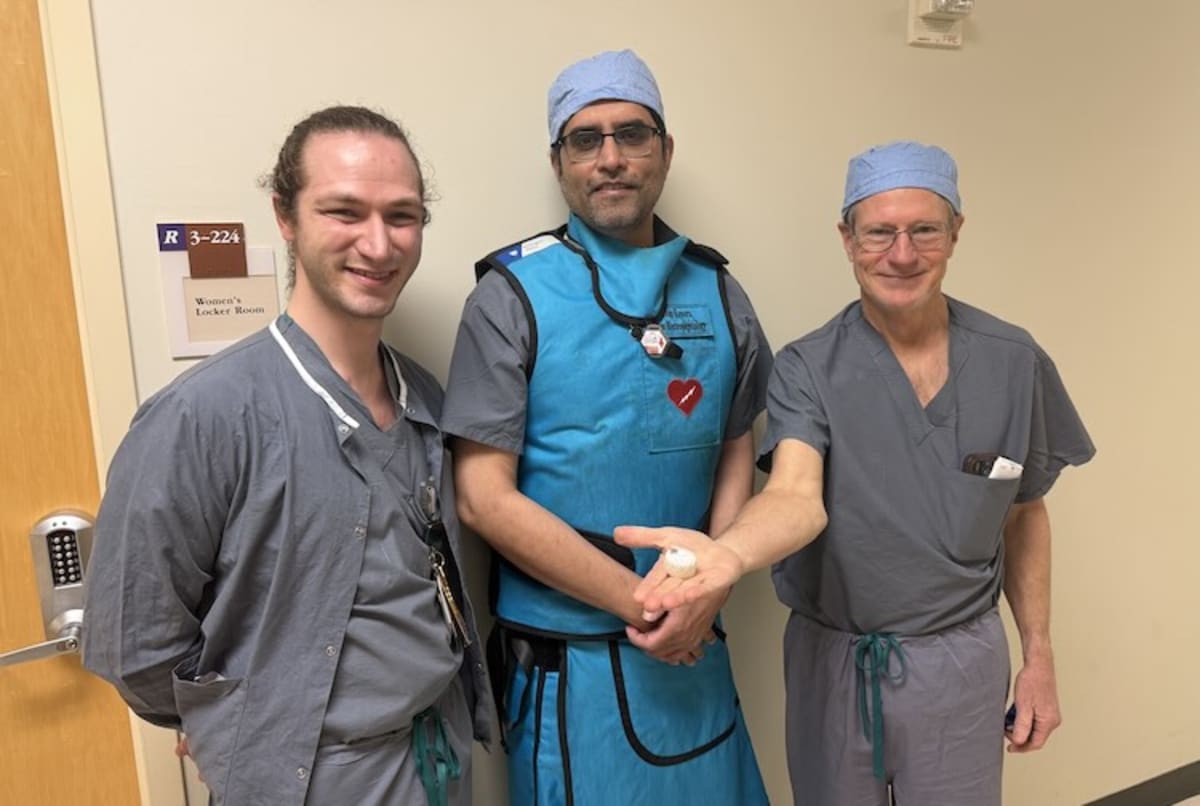
VCU Health Pauley Heart Center recently became the first in Virginia to implant the CLAAS AcuFORM device in a patient with atrial fibrillation, the most common heart rhythm disorder affecting over 10 million Americans. The condition causes blood to pool in a small pocket of the heart, where it can form dangerous clots that lead to stroke.
The new device works by sealing off that pocket, called the left atrial appendage, using a special foam that molds to each patient's unique heart shape. This matters because existing devices don't work for everyone, leaving some patients without good options.
"Blood thinners can help prevent strokes in people living with AFib, but some patients either can't or don't want to take these medications," said Dr. Greg Hundley, director of VCU Health Pauley Heart Center. For those patients, this procedure offers a promising alternative.
The team performed their first implant on February 10 as part of a nationwide clinical trial comparing the foam device to other stroke prevention options. The trial will help determine if the flexible foam design can successfully protect patients with more complex heart anatomies who haven't had good treatment choices before.

The timing couldn't be better. AFib affects an estimated 10 million Americans today, and that number is expected to climb to 12 million by 2030. Between 10% and 20% of all strokes are caused by the condition.
The Ripple Effect
VCU Health Pauley Heart Center has been a pioneer in heart rhythm treatments for over 30 years. They were among the first three hospitals in the entire United States to perform catheter ablation for AFib back in 1998, a procedure that became the standard treatment for the condition.
The center also participated in the original trials for the WATCHMAN device, which became the first FDA-approved left atrial appendage closure device in 2015. Their continued involvement in cutting-edge research means Virginia patients get access to potentially life-saving treatments years before they become widely available.
Dr. Jayanthi Koneru and Dr. Richard Shepard, the electrophysiologists who performed the first Virginia implant, are helping pave the way for more personalized heart care. Their work could eventually give doctors better tools to protect patients with all types of heart anatomy.
As the trial continues, the hope is simple: fewer strokes, more lives saved, and better options for everyone.
Based on reporting by Google News - New Treatment
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it