
Scientists Stop Catalysts from Losing Power with 1 Atom
Researchers discovered that adding just one platinum atom per hundred copper atoms keeps industrial catalysts working at full strength. This breakthrough could make manufacturing everything from medicine to fuel more efficient and sustainable.
Scientists just figured out how to keep one of industry's most important tools from wearing out, and the solution is surprisingly simple.
Researchers at Stanford and partner institutions discovered that adding a tiny amount of platinum to copper catalysts prevents them from degrading during high-temperature manufacturing processes. These catalysts speed up production of plastics, fertilizers, medicines, and fuels by a thousandfold, but they've always had a frustrating weakness.
The problem is called sintering. When temperatures rise during manufacturing, copper atoms start jumping around like popcorn kernels, clustering together into larger particles. This doubles or triples the particle size, shrinking the active surface area and grinding production to a crawl.
For years, manufacturers whispered about a possible fix: adding trace amounts of other metals seemed to help. But nobody understood why, so companies were stuck playing an expensive guessing game with every new catalyst design.
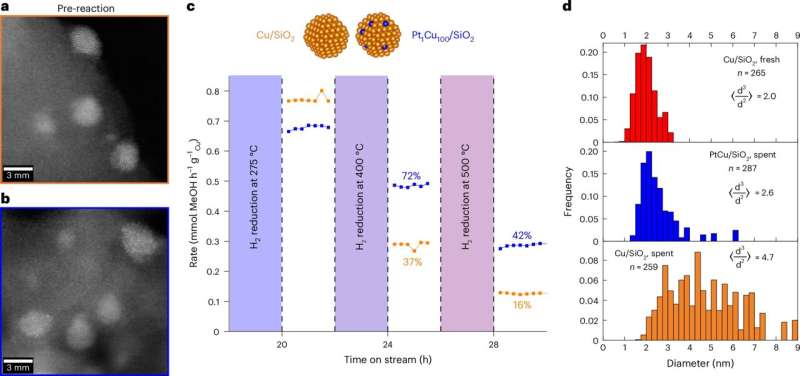
The research team tested copper samples with and without platinum, using three different scientific approaches. At UC Santa Barbara, they aged the catalysts rapidly and found that regular copper lost 84% of its power while platinum-enhanced copper kept 42% of its strength.

At Tufts University, researchers watched individual atoms through powerful microscopes as temperatures climbed. Regular copper atoms hopped around wildly, but platinum-treated copper stayed remarkably stable.
The Stanford team used X-rays to watch the catalysts work in real time under actual manufacturing conditions. Regular copper particles ballooned from less than 1 nanometer to over 3 nanometers, losing 80% of their active surface area. The platinum-enhanced version stayed under 1 nanometer.
The breakthrough came when they figured out the mechanism. Platinum atoms anchor themselves to vulnerable spots on the copper particles called edge and kink sites, blocking copper atoms from escaping and merging with larger particles. It's like putting tiny locks on the exact doors atoms use to leave.
Why This Inspires
The elegance of this solution is stunning. Just one platinum atom per hundred copper atoms makes the difference between a catalyst that degrades quickly and one that maintains its power. That's less than 1% of an additive creating massive improvements in efficiency.
This discovery means manufacturers can now design better catalysts on purpose instead of through trial and error. Simon Bare, who leads the Chemistry & Catalysis Division at Stanford, emphasized that understanding the fundamental mechanism unlocks the ability to improve countless other industrial processes.
The implications ripple far beyond copper and platinum. Chemical manufacturing underpins modern life, from the fertilizers that feed billions to the medicines that save lives. More efficient catalysts mean lower energy costs, less waste, and smaller environmental footprints across entire industries.
Better catalysts working longer means cleaner manufacturing for everything we depend on.
More Images


Based on reporting by Phys.org
This story was written by BrightWire based on verified news reports.
Spread the positivity!
Share this good news with someone who needs it


